Building evidence-based medicine skills in gynecology
Deciphering the evidence, and emerging with the best management approach, requires knowing the basics of study design and interpretation as well as the intricacies of patient interaction. Here, a primer.
In thIS article
- RCTs: The good, the bad, and the ugly
- Systematic reviews: What, why, and how?
- Assessing harms
- Applying the evidence and your expertise to your patient
Although evidence-based medicine, or EBM, is not a new concept, the phrase is tossed about frequently in today’s culture of quality improvement initiatives and metrics. What does EBM really mean, however, and how do we ensure we are practicing it?
At its heart, EBM integrates 3 components:
- the individual clinician’s expertise
- the patient’s values and preferences
- the best external evidence to guide treatment decisions.
Because each clinician’s skillset and each patient’s issues and preferences may be quite varied, in this article we target the third piece—determining the best external evidence.
Our focus on EBM is not meant to negate the importance of the clinician’s expertise, which has been gained through years of practice. Indeed, without expertise, “practice risks becoming tyrannized by evidence.”1 However, without current best evidence, “practice risks becoming rapidly out of date, to the detriment of patients.”1 With the integration of evidence, expertise, and patient choice, EBM is not “cookbook” medicine, and it is not conducted only from armchairs and ivory towers. Rather, EBM is, or should be, at the frontline of clinical care.
EBM begins with a specific clinical question, such as “What is the best treatment option for my patient?” The answer can be honed with the “PICO” approach, which considers Population, Intervention, Comparators, and Outcomes of interest. Specifically, in a particular patient population (similar to your own patient), how does an intervention impact key outcomes?
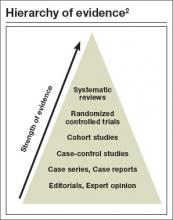
For directly comparing intervention options, such as surgery A versus surgery B, a randomized controlled trial (RCT) is one of the best methods to address clinical questions (FIGURE).2 Systematic reviews are more generalizable than single studies since they compare a range of relevant interventions across populations and settings. Evaluations of diagnostic test accuracy3,4or analyses of risk factors or natural history are best addressed by other study designs, which also can provide important evidence, but will not be discussed in depth here.
In this article, we focus on the benefits of RCTs and systematic reviews, as well as when to exhibit caution, for instance when RCTs report “surrogate outcomes” or make analyses drawn from subgroups of the original population. In addition, we discuss the inability to adequately assess treatment harms (versus benefits) from available evidence as well as the practicalities of how to apply EBM to patients.
RCTs: The good, the bad, and the ugly
RCTs are prospective experiments with a predefined protocol in which patients are randomly allocated to groups where the only difference is the intervention (vs comparators). This design helps to minimize the effects of known and unknown confounders and selection bias.
Ideally, the group into which a study participant is allocated is concealed from the patient and from the caregiver, minimizing the risk that the randomization is broken and the treatment allocation is biased. (Frequently this is not possible, however, particularly for surgical interventions.) Similarly, ideally, the outcome assessors are blinded to the treatment whenever possible. This minimizes the risk of a patient’s outcome being consciously or unconsciously altered due to the outcome assessor’s beliefs about the effectiveness of the intervention.
The reported clinical or surrogate outcomes (which will be discussed in more depth on the next page) for an RCT may be objective or subjective. Preferably, outcomes are patient-centered—important from the patient’s perspective of benefits and harms. Examples of these types of outcomes include survival, function, symptoms, and health-related quality of life, as well as impact on work and family, convenience, and cost. Patients likely are less interested in estimated blood loss, surgical time, biochemistry results, and other clinical or surrogate outcomes.
There are disadvantages to RCTs. For instance, each study provides only a snapshot of the evidence on a given topic. One study rarely, if ever, provides a definitive conclusion. The study’s findings are subject to random error and to biases introduced by study design or analytic methods, and they will not be generalizable to all patients and settings. In addition, the study likely has evaluated only 1 or 2 specific interventions among a plethora of available options, and is unlikely to have analyzed all outcomes of interest.
It becomes your burden to assess whether a trial’s findings are applicable to an actual patient (known as “external validity”). Because an RCT must artificially constrain the underlying clinical questions into a testable research question, translation to the specific patient is often flawed. Perhaps the patient does not precisely fit the inclusion criteria of the trial, for instance, or the exact intervention tested is not fully reproducible. From a practicality perspective, an RCT is often immensely costly to execute, which may be reflected in relatively small numbers of patients and short-term duration of follow-up. These disadvantages limit the ability of RCTs to assess harms, rare events, and long-term outcomes.