2015 Update on cervical disease: New ammo for HPV prevention and screening
Every cervical cancer begins with human papillomavirus (HPV) infection. A new vaccine expands protection against this infection, and a new HPV test offers an effective and efficient screening strategy for women 25 years and older.
In this article
- Disease progression with 9-valent versus quadrivalent HPV vaccine
- How well do current screening strategies detect disease?
- Recommended primary HPV screening algorithm
Two very recent significant advances in cervical disease prevention and screening make this an exciting time for women’s health clinicians. One development, the 9-valent human papillomavirus (HPV) vaccine, offers the potential to increase overall prevention of cervical cancer to over 90%. The other advance offers clinicians a cervical cancer screening alternative, HPV DNA testing, for primary cervical cancer screening. In this article, I underscore the data behind, as well as expert guidance on, these two important developments.
The 9-valent HPV vaccine expands HPV-type coverage and vaccine options for routine use
Joura EA, Giuliano AR, Iversen O, et al. A 9-valent vaccine against infection and intraepithelial neoplasia in women. N Engl J Med. 2015;372(8):711–723.
Two HPV types, 16 and 18, cause the majority—about 70%—of cervical cancers. Vaccination against these types, as well as against types 6 and 11 that cause most condyloma, has been available in the United States since 2006, when the quadrivalent vaccine was approved by the US Food and Drug Administration (FDA).1 Now, based on the results of Joura and colleagues’ randomized, double-blind phase 2b−3 study involving more than 14,000 women, the 9-valent vaccine (Gardasil 9, Merck, Whitehouse Station, New Jersey) has been recommended by the Advisory Committee on Immunization Practices (ACIP) as 1 of 3 HPV vaccines that can be used for routine vaccination.1 (The other 2 vaccines include the bivalent [Cervarix, GlaxoSmithKline, Research Triangle Park, North Carolina] and quadrivalent [Gardasil, Merck]).
Compared with quadrivalent, does the 9-valent vaccine offer compelling additional protection?
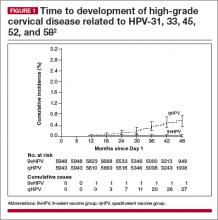
The incidence rate of high-grade cervical intraepithelial neoplasia (CIN; ≥CIN 2 or adenocarcinoma in situ) related to the additional HPV types covered with the 9-valent vaccine (31, 33, 45, 52, and 58) was 0.1 per 1,000 person-years in the 9-valent group and 1.6 per 1,000 person-years in the quadrivalent group. This is equivalent to 1 case versus 30 cases of disease and translates to 96.7% efficacy (95% confidence interval [CI], 80.9−99.8) against these 5 additional high-risk HPV types. At 36 months, there was 1 case of high-grade cervical disease in the 9-valent group related to the 5 additional HPV types, compared with 20 cumulative cases in the quadrivalent group. At 48 months, there was 1 case in the 9-valent group and 27 cases in the quadrivalent group (FIGURE 1).
This expanded disease coverage means the vaccine has the potential to prevent an additional 15% to 20% of cervical cancers in addition to the potential to prevent 5% to 20% of other HPV-related cancers.3
The added HPV-type protection resulted in more frequent injection site reactions (90.7% in the 9-valent group vs 84.9% in the quadrivalent group). Pain, erythema, and pruritis were the most common reactions. While rare, events of severe intensity were more common in the 9-valent group. However, less than 0.1% of participants discontinued study vaccination because of a vaccine-related adverse event.
Study strengths and weaknesses
This was a well-designed prospective, randomized controlled trial. Follow-up was limited; however, this is typical for a clinical trial, and extended follow-up analyses have held up in other HPV vaccine trials; I don’t anticipate it will be any different in this case. The control arm in the case of this trial was the quadrivalent vaccine, as that is the routinely recommended vaccine, so it is not ethical to give placebo in this age-range population. The placebo study already was published,4 so Joura and colleagues’ results build on prior findings.
What this EVIDENCE means for practice
In a widely vaccinated population, the 9-valent HPV vaccine has the potential to protect against an additional 20% of cervical cancers, compared with the quadrivalent vaccine. This is an important improvement in HPV infection and cervical disease prevention. Unfortunately, in the United States we still have very low coverage for the first dose of the HPV vaccine, and even lower coverage for the recommended 3-dose series. This is a big problem in the United States. Stakeholders and advocates need to figure out innovative ways to overcome the challenges of full vaccination for the patients in whom it’s routinely recommended—11- and 12-year-old girls and boys. HPV vaccination lags behind coverage for other vaccines recommended in this same age group—by 20% to 25%.3 US HPV vaccination rates are woefully low in comparison with such other countries as Australia, much of western Europe, and the UK. “If teenagers were offered and accepted HPV vaccination every time they received another vaccine, first-dose coverage for HPV would exceed 90%.”3
The ACIP recommends routine vaccination for HPV—with the bivalent, quadrivalent, or 9-valent vaccine—at age 11 or 12 years. They also recommend vaccination for females aged 13 through 26 years and males aged 13 through 21 years who have not been vaccinated previously. Vaccination is also recommended through age 26 years for men who have sex with men and for immunocompromised persons (including those with HIV infection) if not vaccinated previously.1
By the time I retire, I hope that the impact of protection against additional HPV infection types will be felt, with HPV vaccination rates improved and fewer women affected by the morbidity and mortality related to cervical cancer. As ObGyns, we want to do right by our patients; we need to embrace and continue to discuss the message of primary protection with vaccines that protect against HPV in order to overcome the mixed rhetoric patients and parents receive from other groups, including sensational media or political figureheads who might have an alternative agenda that is clearly not in the best interest of our patients.
HPV test alone is as effective as Pap plus HPV test for cervical disease screening
Wright TC, Stoler MH, Behrens CM, Sharma A, Zhang G, Wright TL. Primary cervical cancer screening with human papillomavirus: end of study results from the ATHENA study using HPV as the first-line screening test. Gynecol Oncol. 2015;136(2):189–197.
The cobas (Roche Molecular Diagnostics, Pleasanton, California) HPV DNA test received FDA approval as a primary screening test for cervical cancer in women aged 25 and older in April 2015. This is a big paradigm shift from what has long been the way we screen women, starting with cytology. Simplistically, the thinking is that we start with the more sensitive test to enrich the population of women that might need additional testing, which might include cytology.
The FDA considered these end-of-study data by Wright and colleagues, which had not been publically published at the time, in its decision. With the Addressing the Need for Advanced HPV Diagnostics (ATHENA) 3-year prospective study, these investigators sought to address major unresolved issues related to HPV primary screening, such as determining which HPV-positive women should be referred to colposcopy and how HPV primary screening performs in the United States. Such a strategy long has been shown to be effective in large prospective European trials.
Details of the study
Three screening strategies were tested:
- Cytology: HPV testing performed only for atypical cells of undetermined significance (ASC-US).
- Hybrid: Cytology strategy for women aged 25 to 29 and cotesting with both cytology and HPV (pooled 14 genotypes) for women 30 years or older. This strategy mimics current preferred US screening recommendations. With cotesting, HPV-positive women with negative cytology are retested with both tests in 1 year and undergo colposcopy if either test is abnormal.
- HPV primary: HPV-negative women rescreened in 3 years, HPV16/18-positive women receive immediate colposcopy, women positive for the other 12 HPV types receive reflex cytology with colposcopy if the cytology is ASC-US or worse. If cytology results are negative, women are rescreened with HPV and cytology in 1 year.
In all strategies, women who were referred to colposcopy and found not to have CIN 2 or greater were rescreened with both tests in 1 year and referred to colposcopy if the finding was ASC-US or higher-grade or persistently HPV-positive.
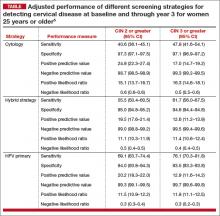
Of the 3 screening strategies, HPV primary in women 25 years and older had the highest adjusted sensitivity over 3 years (76.1%; 95% CI, 70.3–81.8) for the detection of CIN 3 or greater, with similar specificity as the cytology and hybrid strategies. In addition, the negative predictive value for not having clinically relevant disease for HPV primary was comparable to or better than the other 2 strategies (TABLE).5
Another important finding was that the number of colposcopies required to detect 1 case of cervical disease, although found to be significantly higher, was comparable for the HPV primary and cytology strategies (7.1 [95% CI, 6.4–8.0] for cytology vs 8.0 for HPV primary for CIN 2 or greater in women 25 years and older). For CIN 3 or greater, the number of colposcopies required to detect 1 case was 12.8 (95% CI, 11.7–14.5) for HPV primary versus 12.9 (95% CI, 11.5–14.8) for hybrid and 10.8 (95% CI, 9.4–12.6) for cytology.
What this EVIDENCE means for practice
These data indicate that HPV primary screening in women aged 25 and older is as effective as a hybrid screening strategy that uses cytology and cotesting in a patient older than 30 years. And HPV primary screening requires fewer overall screening tests to identify women who have clinically significant cervical disease.
Importantly, compared with a cytology-based strategy, the negative predictive value is quite high for HPV primary screening. Therefore, if someone has a negative HPV test result, the likelihood of that person actually having some sort of clinically relevant disease that day or in the next 3 years is incredibly low. And this is really what’s important for our patients who are getting screened for cervical cancer.