Endometrial Cancer Update: The move toward personalized cancer care
Endometrial cancer is the most common malignancy of the female reproductive tract in the United States, and its incidence continues to rise, with an estimated 49,560 new cases predicted for 2013.1 If we are to successfully traverse the pathway from molecular cell genetics to the development of targeted therapies and personalized cancer care, we need to meet a few benchmarks:
- We need to enhance our understanding of the molecular changes that lead to endometrial cancer. Of particular interest are nonendometrioid tumors. Increased mortality from endometrial cancer appears to be related to the growing number of uterine papillary serous carcinomas and clear-cell cancers. Although these cancers constitute less than 10% of all endometrial cancers, they account for a disproportionately high number of recurrences and cancer-related deaths.2,3 Do recent studies validate the original classification of endometrial cancers as Type I (endometrioid) or Type II (serous and clear cell), or is there more heterogeneity than was originally thought? How do recent studies affect treatment options?
- We need to establish a genomic characterization of endometrial cancer to supplement clinical research data. The identification of novel mutations specific to each histologic type has the potential to improve adjuvant therapy. How close are we to performing a comprehensive genomic analysis of endometrial cancer?
- We need to develop new adjuvant treatment options for recurrent and advanced disease. When clinical symptoms of endometrial cancer are overt, as they often are, early diagnosis is possible, with a 5-year survival rate of 80% to 90%. The prognosis declines dramatically in women with advanced-stage disease or high-risk histologies, with a 5-year survival rate of 57% and 19% for Stage III and Stage IV disease, respectively.1 Adjuvant treatment options are limited in the setting of recurrent or advanced disease. Do any biologic agents increase survival?
In this article, we highlight the historical foundation and newest advances in the field of endometrial cancer, focusing on:
- histologic classification
- etiologic heterogeneity and molecular biology
- genome-guided clinical trials involving targeted therapy, with the ultimate goal of achieving individualized cancer care.
Should we reclassify endometrial cancers to reflect molecular characteristics of tumors?
Brinton LA, Felix AS, McMeekin DS, et al. Etiologic heterogeneity in endometrial cancer: evidence from a Gynecologic Oncology Group trial. Gynecol Oncol. 2013;129(2):277–284.
Cancer Genome Atlas Research Network, et al. Integrated genomic characterization of endometrial carcinoma. Nature. 2013;497(7447):67–73.
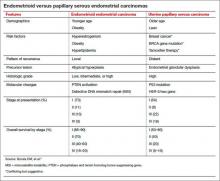
Uterine papillary serous carcinoma (UPSC) was first established as a distinct subtype of endometrial cancer in the early 1980s, when teams led by Lauchlan4 and Hendrickson5 described it as histologically similar to serous epithelial ovarian carcinoma. Shortly thereafter, Bokhman proposed two broad categories of endometrial carcinoma characterized by distinct microscopic appearance, epidemiology, and endocrine and metabolic functioning (TABLE, page 28).6
More recently, research has focused on expanding this histologic classification system to encompass molecular differences. Brinton and colleagues conducted a study within Gynecologic Oncology Group 210, investigating the etiologic heterogeneity of endometrial cancers by comparing risk factors for different histologies. They found that risk factors for aggressive endometrial cancers, including Grade 3 endometrioid and nonendometrioid tumors, appear to differ from those of lower-grade endometrioid carcinomas.
Details of the study by Brinton and colleagues
A total of 3,434 women were included, representing endometrioid (78%) and serous (9%) carcinomas. Grade 3 endometrioid tumors resembled Type II endometrial cancers more closely than did Grade 1–2 endometrioid tumors. Patients with Grade 3 endometrioid and Type II cancers were diagnosed at a significantly older age than patients with Grades 1–2 endometrioid cancers (eg, diagnosis of serous cancers: median age, 67.4 years; Grade 3 endometrioid cancers: median age, 61.9 years; Grade 1–2 endometrioid cancers: median age, 59.6 years). They also were more likely to be nonwhite than patients with Grades 1–2 endometrioid histology. Specifically, black patients were rarely diagnosed with Grades 1–2 endometrioid cancers (5% vs 9% for Grade 3 endometrioid cancers; 20% for serous cancers, 23% for carcinosarcomas, and 12% for clear-cell cancers).
After adjustments for age, enrollment year, and race, patients with Type II tumors (serous, carcinosarcomas, or clear-cell tumors) were much more likely to be multiparous or smokers or to have a history of breast cancer treated with tamoxifen, compared with women with Grade 1–2 endometrioid cancers. An adequately powered subanalysis of serous carcinomas and Grades 1–2 endometrioid cancers revealed that associations persisted between serous carcinomas and multiparity, body mass index, and a history of breast cancer treated with tamoxifen.
Related article: The future of the Pap test: Identifying endometrial and ovarian cancers (Janelle Yates, August 2013)
Overall, this study provides some of the strongest epidemiologic support we have that endometrial cancers are heterogeneous, with evidence to suggest that we might classify Grade 3 endometrioid carcinomas as Type II cancers. These findings paved the way for molecular profiling of endometrial cancers.