Feeding during High-Flow Nasal Cannula for Bronchiolitis: Associations with Time to Discharge
BACKGROUND: High-flow nasal cannula (HFNC) is increasingly used to treat children hospitalized with bronchiolitis; however, the best practices for feeding during HFNC and the impact of feeding on time to discharge and adverse events are unknown. The study objective was to assess whether feeding exposure during HFNC was associated with time to discharge or feeding-related adverse events.
METHODS: This retrospective cohort study included inpatients aged 1-24 months receiving HFNC for bronchiolitis at an academic children’s hospital from January 1, 2015 to March 1, 2017. Feeding exposures during HFNC were categorized as fed or not fed. Among fed children, we further evaluated mixed (oral and tube) or exclusive oral feeding. The primary outcome was time to discharge after HFNC cessation. Secondary outcomes were aspiration, intubation after HFNC, and seven-day readmission.
RESULTS: Of 123 children treated with HFNC, 45 (37 %) were never fed. A total of 78 children (63%) were fed; 50 (41%) were exclusively orally fed and 28 (23 %) had mixed feeding. Median (interquartile range) time to discharge after HFNC was 29.5 hours (23.5-47.9) and 39.8 hours (26.4-61.5) hours in the fed and not fed groups, respectively. In adjusted models, time to discharge was shorter with any feeding (hazard ratio [HR] 2.17; 95% CI: 1.34-3.50) and with exclusive oral feeding (HR 2.13; 95% CI: 1.31-3.45) compared with no feeding. Time to discharge from HFNC initiation was shorter for exclusive oral feeding versus not feeding (propensity weighted HR 1.97 [95% CI: 1.13-3.43]). Adverse events (one intubation, one aspiration pneumonia, one readmission) occurred in both groups.
LIMITATIONS: Assessment of feeding exposure did not account for quantity and duration.
DISCUSSION: Children fed while receiving HFNC for bronchiolitis may have shorter time to discharge than those not fed. Feeding-related adverse events were rare regardless of the feeding method. Controlled prospective studies addressing residual confounding are needed to justify a change in the current practice.
© 2019 Society of Hospital Medicine
Statistical Analysis
Descriptive statistics evaluated differences in demographics and clinical variables for feeding exposure groups. We used chi-squared tests for differences in proportions and t-tests or Wilcoxon Rank-Sum tests for differences in means or medians for continuous variables, respectively. Associations between feeding exposure during HFNC and time to discharge (measured in hours) after HFNC completion were modeled with Cox proportional hazards regression. Using this approach, hazard ratios (HR)>1 indicate a higher hazard (rate) of discharge for children with a feeding exposure than for children without the exposure. For example, a hazard ratio equal to two indicates that the exposed population is discharged at twice the rate per unit time as the nonexposed population. Death or censoring events did not occur. Feeding exposure was first modeled dichotomously as not fed or fed. To further explore associations between feeding modality and our outcome, we then modeled feeding exposure categorically as not fed (reference), mixed (oral and tube) feeding, or exclusive oral feeding throughout HFNC.
After constructing a set of unadjusted models, we then adjusted the models for variables having independent (bivariate P < .10) associations with time to discharge: age, unit of HFNC initiation, highest respiratory support required before HFNC, and HFNC duration. Finally, to attempt to account for residual confounding from latent constructs, we also created a set of propensity-weighted Cox proportional hazards models. Propensity weights18 reflecting the probability of being fed or never being fed during HFNC were created using logistic regression with predictors we hypothesized a priori that may have influenced the clinical decision to feed during HFNC: age, day of illness on admission, prematurity, PIM2 ROM score, respiratory rate, oxygen saturation and blood gas acquisition at HFNC initiation, and highest respiratory support required before HFNC. All analyses were conducted using STATA 14.0 (StataCorp, College Station, Texas), and adjusted hazard ratios (aHR) with 95% confidence intervals (95% CIs) were reported.
RESULTS
Patients (n = 123) had a mean age of 7.3 months (standard deviation [SD] 7.1) and presented on day of illness 4.8 (SD 2.3). Prior to HFNC, 10% required higher respiratory support (3% mechanical ventilation). Former preterm children were 12% of the overall sample.
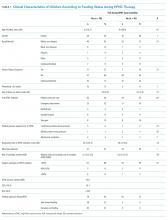
During HFNC, 37% of patients were never fed, 41% were exclusively orally fed, and 23% had tube or mixed oral and tube feedings (Table 1 and Appendix 2). Children who were not fed were older, but groups were otherwise similar in terms of gender, race/ethnicity, passive smoke exposure, day of illness, unit of HFNC initiation, respiratory support required prior to HFNC, and respiratory rate at HFNC initiation.
Median time to discharge after HFNC completion was 31.4 hours (interquartile range [IQR] 23.9-52). Median (IQR) time to discharge was 29.5 (IQR 23.5-47.9) hours in children who were fed and 39.8 (26.4-61.5) hours in those who were not fed (unadjusted HR 1.25 [0.86-1.82], aHR 1.83 [95% CI: 1.16-2.88]). Time to discharge was shortest when children were fed exclusively orally (Figure). Compared with children who were not fed, time to discharge following HFNC completion was significantly shorter for those who were exclusively orally fed (aHR 2.13 [95% CI: 1.31-3.45]; Table 2). Results of the propensity-weighted model were similar: time to discharge after completing HFNC was shorter in fed versus not fed children (HR 2.17; 95 % CI: 1.34-3.50). The secondary outcome, time to discharge from HFNC initiation, had a similar relationship, ie, shorter time to discharge with exclusive oral feeding vs not feeding [aHR 1.95 (95% CI: 1.19-3.18)]. Time to discharge after initiation of HFNC was also shorter for fed versus not fed in propensity-weighted analysis (HR 1.97; 95% CI: 1.13-3.43).