Things We Do for No Reason: Intermittent Pneumatic Compression for Medical Ward Patients?
Journal of Hospital Medicine 14(1). 2019 January;47-50 | 10.12788/jhm.3114
January 8, 2019|Journal of Hospital Medicine
© 2019 Society of Hospital Medicine
WHAT SHOULD WE DO INSTEAD?
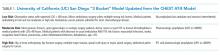
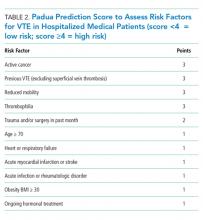
First, one should consider if VTE prophylaxis is needed based on risk assessment. According to the Agency for Healthcare Research and Quality (AHRQ), the most widely used risk stratification model is the University of California San Diego “3 bucket model” (Table 1) derived from tables in ACCP-AT8 guidelines.31The Caprini risk assessment model has been validated for surgical patients, but AHRQ offers caveats related to the complexity of the tool, the difficulty many sites have integrating it into order sets, and the negative experience of the Michigan Hospital Medicine Safety Consortium. The consortium enrolled 43 hospitals with the great majority using the Caprini risk assessment model, but it failed to reduce VTE in medical patients.31 Alternatively, the ACCP-AT9 guidelines recommend the Padua prediction score for risk assessment of medical patients (Table 2). VTE occurs in 0.3% of low-risk patients (Padua score <4) and 11.0% of high-risk patients (Padua score ≥4). If IPC is used in the low-risk populations with a predicted VTE rate of 0.3, then 666 patients would need to be treated to prevent one VTE. Treating 666 patients would cost $79,920 for IPC sleeves alone plus $5,500-$7,500 per pump and result in 20 additional episodes of skin breakdown. Therefore, IPC should be reserved for high-risk populations with contraindications to pharmacologic prophylaxis.
- The VTE risk of general medicine ward patients should be assessed, preferably with the “3 bucket” or Padua risk assessment models.
- For low-risk patients, no VTE prophylaxis is indicated. Ambulation ought to be encouraged for low-risk patients.
- If prophylaxis is indicated, then bleeding risk should be assessed to determine a contraindication to pharmacologic prophylaxis. If there is excessive bleeding risk, then treatment with IPC may be considered even though there are only data to support this in high-risk populations such as surgical, stroke, trauma, and critical care patients.
- If using IPC, then strategies that ensure compliance and consider patient comfort based on type and location of sleeves should be implemented.
- Combined IPC and pharmacologic prophylaxis should be used for high-risk trauma or surgical patients.
No current evidence supports IPC efficacy in general medical ward patients despite its widespread use; thus, prospective trials in this population are needed. Given costs, potential side effects, and uncertain efficacy in general medical ward patients, IPC should be reserved for surgical, trauma, critical care, or stroke patients. It may be considered for moderate to high-risk medical patients with excessive bleeding risk. Our clinical scenario patient bled within the past three months (odds ratio for bleeding 3.64; 95% CI, 2.21-5.99).32 On the basis of the increased risk, a dutiful hospitalist might be tempted to order IPC. However, given that our patient is ambulatory, is toileting frequently, and has an expected observation stay of less than 48 hours, he is considered low risk for VTE (Table 1). Additionally, his Padua score of two confirms his low risk status (Table 2). No VTE prophylaxis would be indicated.