Derivation of a Clinical Model to Predict Unchanged Inpatient Echocardiograms
BACKGROUND: Transthoracic echocardiography (TTE) is one of the most commonly ordered tests in healthcare. Repeat TTE, defined as a TTE done within 1 year of a prior TTE, represents 24% to 42% of all studies. The purpose of this study was to derive a clinical prediction model to predict unchanged repeat TTE, with the goal of defining a subset of studies that are unnecessary.
METHODS: Single-center retrospective cohort study of all hospitalized patients who had a repeat TTE between October 1, 2013, and September 30, 2014.
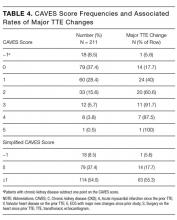
RESULTS: Two hundred eleven of 601 TTEs were repeat studies, of which 78 (37%) had major changes. Five variables were independent predictors of major new TTE changes, including history of intervening acute myocardial infarction, cardiothoracic surgery, major new electrocardiogram (ECG) changes, prior valve disease, and chronic kidney disease. Using the β-coefficient for each of these variables, we defined a clinical prediction model that we named the CAVES score. The acronym CAVES stands for chronic kidney disease, acute myocardial infarction, valvular disease, ECG changes, and surgery (cardiac). The prevalence of major TTE change for the full cohort was 35%. For the group with a CAVES score of −1, that probability was only 5.6%; for the group with a score of 0, the probability was 17.7%; and for the group with a score ≥1, the probability was 55.3%. The bootstrap corrected C statistic for the model was 0.78 (95% confidence interval, 0.72-0.85), indicating good discrimination.
CONCLUSIONS: Overall, the CAVES score had good discrimination and calibration. If further validated, it may be useful to predict repeat TTEs that are unlikely to have major changes.
© 2018 Society of Hospital Medicine
In summary, we have developed a simple score to predict the likelihood of major changes on repeat TTEs for hospitalized patients. The CAVES score identified 8.5% of patients as being low risk for changed repeat TTE, 37% at intermediate risk, and 54% at high risk for major changes. We believe that the CAVES score, if further validated, may be used to risk stratify patients for ordering TTE and to potentially avoid unnecessary repeat studies.
Disclosure
The authors indicated no conflicts of interest.