Small Cell Lung Cancer
INTRODUCTION
Small cell lung cancer (SCLC) is an aggressive cancer of neuroendocrine origin that accounts for approximately 15% of all lung cancer cases, with approximately 33,000 patients diagnosed annually.1 The incidence of SCLC in the United States has steadily declined over the past 30 years, presumably because of a decrease in the number of smokers and a change to low-tar filter cigarettes.2 Although the overall incidence of SCLC has been decreasing, the incidence in women is increasing and the male-to-female incidence ratio is now 1:1.3 Nearly all cases of SCLC are associated with heavy tobacco exposure, making it a heterogeneous disease with a complex genomic landscape consisting of thousands of mutations.4,5 Despite recent advances in the treatment of non-small cell lung cancer, the therapeutic options for SCLC remain limited, with a median overall survival (OS) of 9 months in patients with advanced disease.
DIAGNOSIS AND STAGING
CASE PRESENTATION
A 61-year-old man presents to the emergency department with progressive shortness of breath and cough over the past 6 weeks. He also reports a 20-lb weight loss over the same period. He is a current smoker and has been smoking 1 pack of cigarettes per day since the age of 18 years. A chest radiograph obtained in the emergency department shows a right hilar mass. Computed tomography (CT) scan confirms the presence of a 4.5-cm right hilar mass and enlarged mediastinal lymph nodes bilaterally.
• What are the next steps in diagnosis?
SCLC is characterized by rapid growth and early hematogenous metastasis. Consequently, only 25% of patients have limited-stage disease at the time of diagnosis. According to the Veterans Administration Lung Study Group (VALSG) staging system, limited-stage disease is defined as tumor that is confined to 1 hemithorax and can be encompassed within 1 radiation field. This typically includes mediastinal lymph nodes and ipsilateral supraclavicular lymph nodes. Approximately 75% of patients present with extensive-stage disease, which is defined as disease that cannot be classified as limited, including disease that extends beyond 1 hemithorax. Extensive-stage disease includes the presence of malignant pleural effusion and/or distant metastasis.6 The VALSG classification and staging system is more commonly used in clinical practice than the American Joint Committee on Cancer TNM staging system because it is less complex and directs treatment decisions, as most of the literature on SCLC classifies patients based on the VALSG system.7
Given SCLC’s propensity to metastasize quickly, none of the currently available screening methods have proven successful in early detection of SCLC. In the National Lung Cancer Screening Trial, 86% of the 125 patients who were diagnosed with SCLC while undergoing annual low-dose chest CT scans had advanced disease at diagnosis.8,9 These results highlight the fact that most cases of SCLC develop in the interval between annual screening imaging.
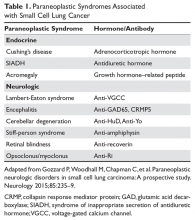
SCLC frequently presents with a large hilar mass that is symptomatic. Common symptoms include shortness of breath and cough. In addition, patients with SCLC usually have bulky mediastinal adenopathy at presentation. SCLC is commonly located submucosally in the bronchus, and therefore hemoptysis is not a very common symptom at the time of presentation. Patients may present with superior vena cava syndrome from local compression by the tumor. Not infrequently, SCLC is associated with paraneoplastic syndromes that arise due to ectopic secretion of hormones or antibodies by the tumor cells. The paraneoplastic syndromes can be broadly categorized as endocrine or neurologic (Table 1). The presence of a paraneoplastic syndrome is often a clue to the potential diagnosis of SCLC in the presence of a hilar mass. Additionally, some paraneoplastic syndromes, more specifically endocrine paraneoplastic syndromes, follow the pattern of disease response and relapse, and therefore can sometimes serve as an early marker of disease relapse or progression.
The common sites of metastases include brain, liver, and bone. Therefore, the staging workup should include fluorodeoxyglucose (FDG) positron emission tomography (PET)/CT scan. Contrast-enhanced CT scan of the chest and abdomen and bone scan can be obtained for staging in lieu of PET scan. Due to the physiologic FDG uptake, cerebral metastases cannot be assessed with sufficient certainty using PET-CT.10 Therefore, brain imaging with contrast-enhanced CT or magnetic resonance imaging (MRI) is also necessary. Although the incidence of metastasis to bone marrow is less than 10%, bone marrow aspiration and biopsy are warranted in patients with unexplained cytopenias, especially when the cytopenia is associated with teardrop-shaped red cells or nucleated red cells on peripheral blood smear, findings indicative of a marrow infiltrative process.7 The tissue diagnosis is established by obtaining a biopsy of the primary tumor or 1 of the metastatic sites. In localized disease, bronchoscopy (with endobronchial ultrasound, if necessary) with biopsy of the centrally located tumor and/or lymph node is required. Histologically, SCLC consists of monomorphic cells, a high nuclear-cytoplasmic ratio, and confluent necrosis. The tumor cells are positive for chromogranin, synaptophysin, and CD56 by immunohistochemistry, and very frequently are also positive for thyroid transcription factor 1.11 Although serum tumor markers, including neuron-specific enolase and progastrin-releasing peptide, are frequently elevated in patients with SCLC, these markers are of limited value in clinical practice because they lack sensitivity and specificity.12