Evaluation of a Pharmacist-Driven Ambulatory Aspirin Deprescribing Protocol
Background: Recent guidelines indicate that aspirin affords less cardiovascular protection and greater bleeding risks in adults aged > 70 years. Deprescribing potentially inappropriate medications is particularly important in older adults, as this population experiences a high risk of adverse effects and polypharmacy. Limited data are available regarding targeted aspirin deprescribing approaches by pharmacists. The objective of this study was to implement and evaluate the success and feasibility of a pharmacist-led aspirin deprescribing protocol for older adults in a primary care setting.
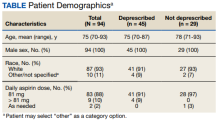
Observations: This prospective feasibility study in a US Department of Veterans Affairs ambulatory care pharmacy setting included patients aged ≥ 70 years with documented aspirin use. We reviewed 459 patient records and determined that 110 were eligible for deprescribing. A pharmacist-initiated telephone call was attempted for each eligible patient to discuss the risks and benefits of deprescribing aspirin. The primary outcome was the proportion of patients reached for whom aspirin was discontinued. Secondary outcomes included patient rationale for declining deprescribing and the time to complete the intervention. Of 94 patients reached, 45 (48%) agreed to aspirin deprescribing, 3 (3%) agreed to dose reduction, and 29 (31%) declined the intervention. An additional 17 (18%) had previously stopped aspirin, which led to a medication reconciliation intervention. Pharmacists spent about 2 minutes per record review and 12 minutes on each encounter, including documentation.
Conclusions: Implementing a pharmacist-driven aspirin deprescribing protocol in a primary care setting led to the discontinuation of inappropriate aspirin prescribing in nearly half of older adults contacted. The protocol was well accepted by collaborating physicians and feasible for pharmacists to implement, with potential for further dissemination across primary care settings.
After identifying eligible patients, a CPP or pharmacy intern contacted patients by telephone, following a script to guide conversation. All patients were screened for potential appropriate aspirin indications, particularly any history of MI, CAD, CVA, TIA, PAD, or other clinical ASCVD. The patient was asked about their rationale for taking aspirin and patient-specific ASCVD risk-enhancing factors and bleeding risk factors and educated them on lifestyle modalities to reduce ASCVD risk, using the script as a guide. ASCVD risk-enhancing factors included family history of premature MI, inability to achieve BP goal, DM with the inability to achieve blood glucose or hemoglobin A1c goal, tobacco use, or inadequate statin therapy. Bleeding risk factors included a history of gastrointestinal bleed or peptic ulcer disease, concurrent use of medications that increase bleeding risk, chronic kidney disease, or thrombocytopenia.
Through shared decision making with careful consideration of these factors, we reached a conclusion with each patient to either continue or to deprescribe aspirin. Each discussion was documented in the electronic health record (EHR) using a standard documentation template (eAppendix, available at doi:10.12788/fp.0320). The patient’s medication list also was updated to reflect changes in aspirin use. For patients who declined deprescribing, the CPP or pharmacy intern asked the patient for their primary reason for preferring to continue aspirin, which was subsequently categorized as one of the following: no prior concerns with bleeding, concerns about a future cardiovascular event, wishing to discuss further with their primary care practitioner (PCP), or identifying an appropriate use for aspirin not evident through record review. For the patients who wished to further discuss the issue with their PCP before deprescribing, the patient’s PCP was notified of this preference by a record alert to the note documenting the encounter, and the patient was also encouraged to follow up about this issue. A voicemail was left if the patient did not answer requesting a call back, and a second attempt was made within 2 weeks.
Data Collected
We collected data to assess the proportion of patients for whom aspirin for primary prevention was discontinued. For patients who declined deprescribing, we documented the rationale for continuing aspirin. Additionally, the feasibility of implementation was assessed, including pharmacist time spent on each record review and intervention. Descriptive statistics were generated to evaluate baseline characteristics and intervention outcomes. The time to completion of these tasks was summarized with descriptive statistics.
We reviewed 459 patient records, and 110 were determined eligible for risk/benefit discussions.
Patients had various reasons for declining deprescribing, including 8 (28%) who had no prior concerns with bleeding while on aspirin and 6 (21%) who were concerned about a future cardiovascular event. Of those who declined aspirin deprescribing, 6 (21%) wished to further discuss the issue with their PCP. In 9 (31%) patients an alternative appropriate indication for aspirin was identified through discussion. In these cases, the indication for aspirin was documented and updated in the EHR.
Most patients (87%) contacted reported taking low-dose aspirin 81 mg daily, while 10% reported taking higher doses (range, 162-325) and 3% on an as-needed basis. In all 3 patients who agreed to dose reduction, the initial dose of 325 mg daily was reduced to 81 mg daily.