Vaginal bowel control device shown safe, effective in pivotal trial
AT AUGS/IUGA 2014
WASHINGTON – Use of a vaginal bowel control device currently under review by the Food and Drug Administration resulted in a significant reduction of fecal incontinence episodes and significant improvement in quality of life, with only mild and transient adverse events, a pivotal study has shown.
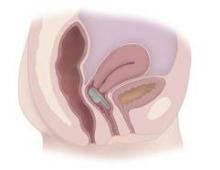
The device – the Vaginal Bowel Control (VBC) System made by Pelvalon (Sunnyvale, Calif.) – consists of a silicone base and a posteriorly directed balloon. The patient controls the inflation pump, inflating the balloon to deflect the rectovaginal septum and interrupt stool passage, and deflating it to allow bowel movements.
"It provides [the patient with] dynamic control of the rectum," said Holly E. Richter, Ph.D., M.D., who served as the national principal investigator of the study. "It’s a new paradigm for treating patients with fecal incontinence."
At the 1-month primary endpoint, 79% of the 61 patients in the study’s intent-to-treat cohort – and 86% of patients in the per-protocol cohort – had experienced treatment success, which was defined as a 50% or greater reduction in fecal incontinence episodes, Dr. Richter reported at the scientific meetings of the American Urogynecologic Society and the International Urogynecological Association.
Mean weekly fecal incontinence episodes were reduced from 5.9 per week at baseline to 1.1 at 1 month.
After the 1-month treatment period, women were given the option of continuing with an additional 2-month study period. The rate of treatment success held steady among the 44 women who continued, with 86% seeing a 50% or greater reduction in fecal incontinence at 3 months, said Dr. Richter, director of the division of urogynecology and pelvic reconstructive surgery at the University of Alabama at Birmingham.
Total continence was achieved in approximately 40% of women in the 1-month and 3-month per-protocol cohorts, she noted.