HIV screening: How we can do better
The CDC and USPSTF recommend routine HIV screening of adults. This review of the tests at your disposal, and a new diagnostic algorithm from the CDC, provide guidance.
› Screen all adolescents and adults ages 15 to 65 years for human immunodeficiency virus (HIV) infection. A
› Screen younger adolescents and older adults who are at increased risk for HIV infection on an annual basis. A
› Screen all pregnant women for HIV infection, including those who are in labor and who are untested or whose HIV status is unknown. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
For the first 15 years of the epidemic, human immunodeficiency virus and acquired immunodeficiency syndrome (HIV/AIDs) was uniformly fatal. Between 1981 and 1996, approximately 362,000 people in the United States succumbed to the disease.1 That began to change in the mid 1990s, though, when highly active antiretroviral therapy (HAART) came into routine use. From that point forward, HIV became a chronic, manageable disease for most patients; an estimated 1.2 million people in the United States are now living with HIV infection.2
Unfortunately, the number of new infections continues to grow. There are more than 50,000 new infections in the United States each year,2 and an estimated approximately 200,000 people have it but are undiagnosed, leading to further spread of the disease.3 The Office of National AIDS Policy has issued a National HIV/AIDS Strategy that seeks to reduce new infections by 25% in 2015, in part by identifying people with the disease who do not know their HIV status.4
But screening still has not gotten the uptake by clinicians that health officials would like.
Lack of awareness by physicians? Or an unwillingness of patients?
In 2006, the Centers for Disease Control and Prevention (CDC) began recommending routine HIV screening for individuals between the ages of 13 and 64, with patients given the ability to opt out of such testing.5 That same year, the CDC also removed some prior barriers to testing, such as requiring written consent and pretest counseling. But as of 2009, fewer than 50% of US adults had ever been tested for HIV6—possibly the result of physicians being unaware of the guidelines, patients being unwilling to be tested, and/or reimbursement issues.
Conflicting recommendations may have played a role. When the CDC released its 2006 recommendations, the United States Preventive Services Task Force (USPSTF) felt there was insufficient evidence to support routine HIV screening and issued a grade C recommendation. At that time, the USPSTF recommended that only high-risk individuals and pregnant women be tested (A recommendation, meaning there was high certainty that the net benefit was substantial).
However, in April 2013, based on new evidence regarding the clinical and public health benefits of early identification of HIV infection and subsequent treatment, the USPSTF updated its recommendations. The USPSTF now encourages clinicians to screen all adolescents and adults age 15 to 65 years for HIV (A recommendation).7 Shortly thereafter, the American Academy of Family Physicians (AAFP) also endorsed routine HIV screening, although the AAFP calls for such screening to begin at age 18.8
Insurance now covers it… A USPSTF A recommendation carries significant health policy implications because the Affordable Care Act requires private and public health insurance plans to cover preventive services recommended by USPSTF.9
Integrating screening into your practice
Serologic tests have come a long way. The first HIV antibody test was an enzyme immunoassay (EIA) that was introduced in 1985 and used mainly to screen the blood supply. This first-generation EIA identified only immunoglobulin G (IgG) antibodies to HIV type 1 (HIV-1). More sensitive and specific second- and third-generation EIAs have since been developed to detect both IgG and IgM antibodies, as well as antibodies to HIV-2. The third-generation assays also can detect antibodies as soon as 3 weeks after infection.
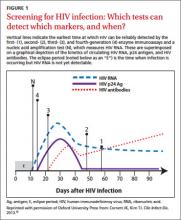
The fourth-generation EIAs were approved by the US Food and Drug Administration (FDA) in 201010 and are the first step in the CDC’s current HIV diagnostic testing algorithm. These tests can detect HIV-1/HIV-2 IgG and IgM antibodies and also p24 antigen, which is present within 7 days of the appearance of HIV RNA.11 The fourth-generation assay allows for reliable detection within about 2 weeks of infection (FIGURE 1).10
Rapid HIV tests are also an option.12 These tests can detect IgG and IgM antibodies in samples of saliva, whole blood, serum, and plasma. Results of rapid tests usually are available in 20 to 30 minutes and allow physicians to give patients the results while they are still in the office. In 2013 the FDA approved a combination p24 antigen/antibody rapid HIV assay that according to the manufacturer can detect infection earlier than other currently available rapid tests.13