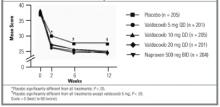
Randomized placebo-controlled trial comparing efficacy and safety of valdecoxib with naproxen in patients with osteoarthritis
Conventional NSAIDs were associated with a significant risk of serious gastrointestinal complications such as ulceration and perforation and low gastrointestinal tolerability.20–22 Naproxen treatment of osteoarthritis and rheumatoid arthritis demonstrated a higher rate of endoscopically proven gastrointestinal ulceration than did COX-2–specific inhibitors,17,23 and that finding was confirmed in this study for 10 mg valdecoxib. Naproxen treatment was associated with significantly more gastroduodenal ulcers than 5 or 10 mg valdecoxib. We found no significant difference between 20 mg valdecoxib and naproxen, which might be explained by a lower incidence of ulcers with naproxen than reported in previous studies.24 In terms of numbers needed to treat, 14 patients would be needed to observe a difference in endoscopic ulcer rates between valdecoxib (5 or 10 mg) and naproxen compared with 20 patients to observe a difference between 20 mg valdecoxib and naproxen and 16 to observe a difference in ulcer rates between naproxen and placebo.
Valdecoxib at a dosage of 10 mg/day also demonstrated overall improved gastrointestinal tolerability, with significantly fewer adverse events and withdrawals due to adverse events, in particular gastrointestinal-related events such as constipation and dyspepsia, than did naproxen. The improved upper gastrointestinal tract safety of valdecoxib was as expected because the COX-1–sparing nature of this agent allows effective inhibition of COX-2 without inhibiting COX-1 in the gastric mucosa and platelets. An improved gastrointestinal safety profile is an important consideration in the treatment of osteoarthritis because the moderate to severe gastrointestinal complications associated with conventional NSAID therapy frequently lead to poor patient compliance or discontinuation of the medication.25,26
Overall, this study suggests clinical benefits of single daily doses of 10 and 20 mg valdecoxib and improved upper gastrointestinal tract safety for the 10-mg dose, compared with 500 mg/day naproxen. No additional efficacy benefit was obtained from a 20-mg dose as opposed to a 10-mg dose. Valdecoxib (10 mg) is a potent and effective once-daily alternative to conventional NSAIDs, with a gastrointestinal safety advantage that will be of value to rheumatologists and primary care physicians alike.