High-Altitude Illness
Patients participating in occupational and sports-related activities requiring ascent to high elevations are at risk of developing a range of high-altitude illnesses. Prompt recognition and treatment are paramount to improving outcomes and preventing life-threatening sequelae.
High-Altitude Cerebral Edema
Mechanism of HACE
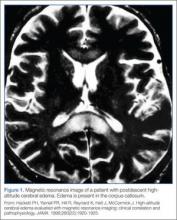
The exact mechanism of HACE is unclear. Magnetic resonance imaging of patients with the condition demonstrates cerebral edema primarily localized to the corpus callosum.11 These findings suggest an increased permeability in the blood-brain barrier, leading to vasogenic cerebral edema. Cases of death associated with HACE are the result of herniation. Fortunately, if the condition is recognized promptly and appropriate management is instituted, most patients will recover without permanent deficits.
Current recommendations for treating HACE are similar to treatment strategies for AMS.
Descent. A therapeutic priority, descent may prove challenging as the patient may be ataxic, have altered mental status, and have difficulty facilitating his or her own descent.
Oxygen. A portable HBO bag can be used to simulate descent until evacuation is possible. Supplemental oxygen should be applied immediately.
Dexamethasone. In treating HACE, dexamethasone may be administered at a loading dose of 8 mg, followed by 4 mg every 6 hours.3
Airway Management. If the patient has significantly altered mental status, appropriate airway management must be initiated.
High-Altitude Pulmonary Edema
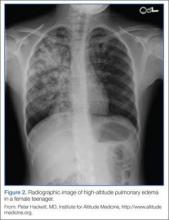
The most common cause of death from altitude illness is HAPE,12 a form of noncardiogenic pulmonary edema. This condition generally occurs at elevations above 3,000 m. Symptoms begin 2 to 5 days after ascent and progress in a typical pattern. A patient will initially experience a nonproductive cough and dyspnea at rest. The dyspnea worsens, and the cough becomes productive of pink, frothy sputum. Without medical intervention, lethargy, coma, and death may follow.
Symptoms of HAPE generally worsen following a night of sleep at elevation. Physical examination reveals crackles, tachycardia, tachypnea, and hypoxia. Diagnosis requires at least two of the following signs:
- Crackles or wheezing in at least one lung field
- Central cyanosis
- Tachypnea
- Tachycardia.
In addition to the above signs, at least two of the following symptoms must also be present:
- Dyspnea at rest
- Cough
- Weakness or decreased exercise performance
- Chest tightness
- Congestion.
Mechanism of HAPE
The mechanism of HAPE is better understood than that of AMS and HACE. In HAPE, high microvascular pressures in the lungs lead to elevated pulmonary vascular resistance and pulmonary artery pressure. Pulmonary edema ensues, but left ventricular function is preserved. Patients with a naturally low HVR, high pulmonary artery pressures at rest, preexisting pulmonary hypertension, or a previous history of HAPE are predisposed to developing the condition. Risk factors include heavy exertion, rapid ascent, cold, salt ingestion, and sleeping medications.
Treatment
Decent and warming of the patient as soon as possible, along with treatment outlined below, are essential.
Oxygen. Treatment of HAPE begins with supplemental oxygen to immediately lower pulmonary artery pressure. Oxygen should initially be administered at 4 to 6 L/min; if the patient improves clinically and can maintain oxygen saturations greater than 90%, oxygen may be decreased with a goal to maintain saturation above 90%.
Nifedipine. Following oxygen, descent, and warming, nifedipine can be used as an adjunctive therapy. The treatment dose for HAPE is 20 to 30 mg of the sustained release form every 12 hours.3
Salmeterol/Albuterol and Expiratory Positive Airway Pressure. The oral inhalers salmeterol or albuterol may be used for bronchodilation; however, there is little evidence to support their effectiveness in HAPE. Ventilation with expiratory positive airway pressure can be employed if available.
Prevention
For patients with a predisposition to HAPE, preventive measures should be considered prior to ascent. As with all forms of altitude illness, gradual ascent is the most effective prevention method available.
Phosphodiesterase Inhibitors. Phosphodiesterase inhibitors act via pulmonary vasodilation to prevent HAPE in some patients. Tadalafil at a dose of 10 mg twice daily or 20 mg once daily has been shown to reduce the incidence of HAPE.14 Alternatively, sildenafil 50 mg three times daily may be used.
Acetazolamide and β-Agonists. Although both acetazolamide and β-agonists such as albuterol have been theorized to aid in preventing HAPE, this has not been proven.15