Lichen Planus and Lichenoid Drug Eruption After Vaccination
Lichen planus (LP) and lichenoid drug eruptions (LDEs) uncommonly occur after vaccination, especially for hepatitis B and influenza. The key initiating event that leads to the development of postimmunization LP or LDE is not well understood. There have been prior reports of an association between several vaccines and LP. In this study, we aim to characterize and review cases of LP and LDE after vaccination from the Vaccine Adverse Event Reporting System (VAERS) national database in the United States. Information on vaccine-associated LP and LDE was retrieved from the database (July 1990 to November 2014) to examine the frequency of LP or LDE after vaccination. Hepatitis B, influenza, and herpes zoster vaccines were the 3 most commonly associated vaccines. Patients with LP or LDE were significantly older compared to the reported adverse events (AEs) overall (P<.001). The median age of onset for LP and LDE was 47 years. The median time of onset of AEs was 14 days. It is important to obtain recent vaccination history in patients presenting with new-onset LP or LDE.
Practice Points
- Lichen planus (LP) and lichenoid drug eruptions (LDEs) can uncommonly occur after vaccination.
- Common vaccines associated with LP and LDEs include hepatitis B and influenza vaccinations.
- It is important to be cognizant of such reactions, especially in patients who have recently received these common vaccines.
Comment
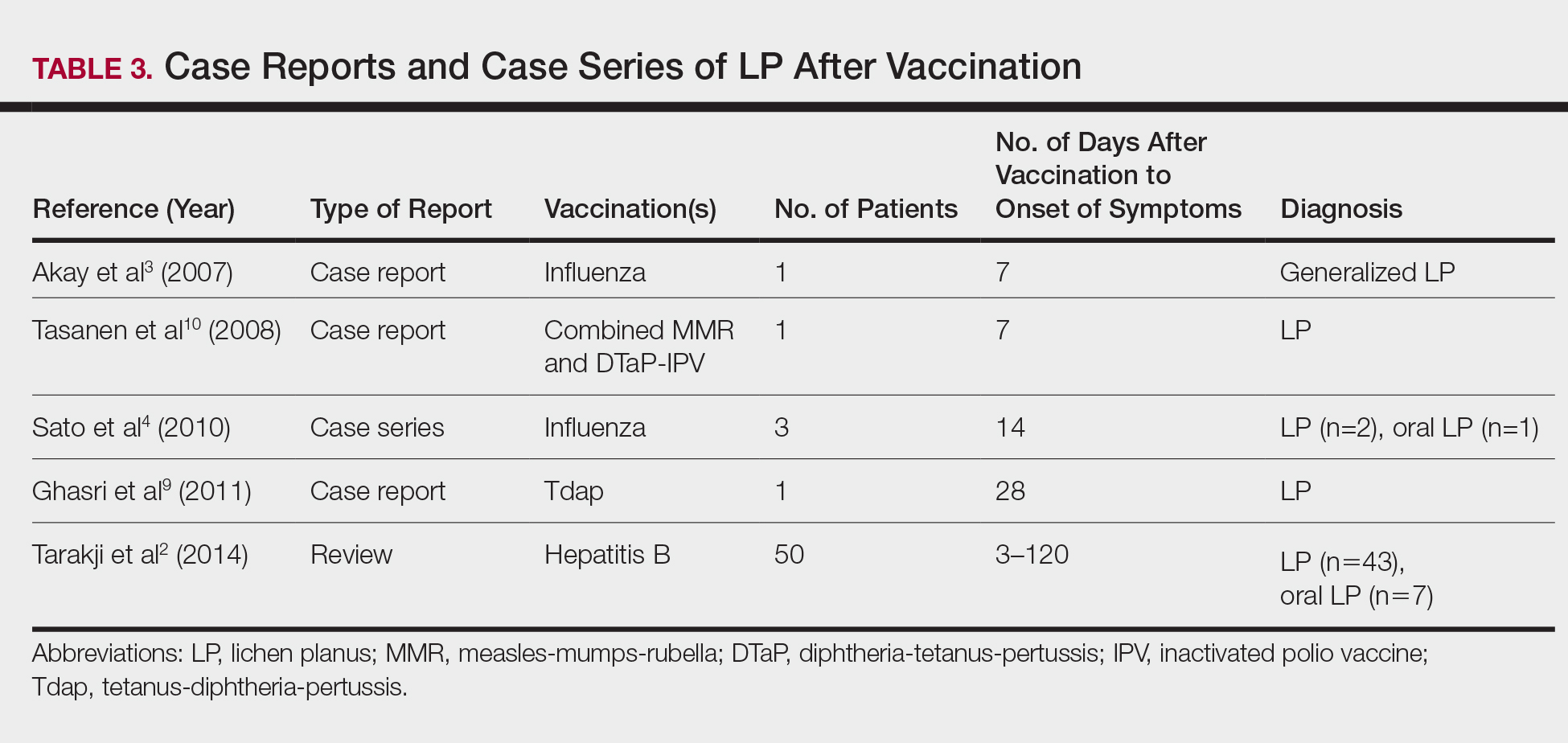
The estimated prevalence of LP ranges from 0.22% to 5% worldwide,11-15 with an incidence of 0.032% to 0.037%.16 Although rare, LP and LDE can occur from certain medications or vaccines. Cases of LP have been reported after hepatitis B and influenza vaccinations. The first case of LP following hepatitis B vaccination was described by Ciaccio and Rebora17 in 1990. Since then, a total of 50 similar cases have been reported worldwide.2 There also have been reports of LP following influenza, tetanus-diphtheria-pertussis, measles-mumps-rubella, and inactivated polio vaccines.3,4,9,10 Table 3 summarizes cases of LP following various vaccinations.
The key initiating event of the pathogenesis for both LP and LDE is not completely understood. Both conditions share similar immunologic mechanisms of persistently activated CD8 autocytotoxic T lymphocytes against epidermal cells.18 These cells can induce apoptosis of basal epidermal keratinocytes and generate various cytokines (eg, IFN-γ, IL-5) to enhance expression of class II MHC molecules and antigen presentation to CD4 T cells.19-22 It is conceivable that one of the initiating factors may be related to components in vaccines.
,Hepatitis B, influenza, and herpes zoster vaccines were the 3 most common vaccines implicated in postimmunization LP or LDEs in our study. The excipients of these vaccines were compared based on the product inserts to identify any common components. It was found that all 3 vaccines contain either yeast protein or egg protein with various forms of phosphate buffers, while the hepatitis A and herpes zoster vaccines share Medical Research Council cell strain 5 (human diploid) cells as well as other cellular components.23 Sato et al4 suggested that specific vaccine components, such as the vaccine itself or egg proteins, could have contributed to the development of LP following vaccination. It has been postulated that the protein S fraction of hepatitis B surface antigen plays a crucial role in the pathogenesis of both LP and LDE after hepatitis B vaccination.2,24 It is likely that protein S shares common epitopes on keratinocytes that are recognized by the immune system, thus activating cytotoxic T lymphocytes and inducing apoptosis.2,24
In this study, the median time to onset of vaccine-related LP was 14 days, which is consistent with a case series by Sato et al,4 suggesting that adverse reactions mainly occurred within 2 weeks after influenza vaccination. Onset of symptoms within 2 weeks of vaccination would therefore be a crucial clue for diagnosing possible vaccine-related LP or LDE. On the other hand, at least 4 patients in our study had onset of LP and LDE more than 1 month after vaccination; 2 of 4 cases even reported symptom onset at 175 and 297 days after hepatitis B vaccination, which were much longer than the 120 days reported by Tarakji et al.2 It is not known if these cases constitute true vaccine-associated LP or LDE or if unmeasured confounding factors such as concurrent medications or comorbidities may have contributed to the development of these AEs.
It also is interesting to note that LP and LDE affected mainly middle-aged women. An increased risk of autoimmunity in female adults partly explains this observation.25 Some vaccines, such as herpes zoster and influenza vaccines, generally are recommended for older adults who also are more likely to have multiple comorbidities or take multiple medications/supplements, which can potentially skew the prevalence of AEs toward an older age group. It should be noted, however, that LP and LDE were relatively uncommon AEs following vaccination in the current study. In this study, LP and LDE consisted of only 0.01% (N=42,230) of all AEs after hepatitis B vaccination, while the more common AEs such as pyrexia, nonspecific rashes, nonspecific gastrointestinal symptoms, and headache contributed to approximately 66.5% of all reported events.
One of the strengths of our study is that up to two-thirds of cases were confirmed histologically and all patients were seen and followed up by dermatologists or physicians. The VAERS is an easily accessible, up-to-date, and live reporting system that collects all AEs associated with vaccines in the United States. Important clinical and laboratory information usually is available in the database; however, the main limitation is that this study can only demonstrate a possible association but not a causal relationship between vaccination and LP or LDE. There can be various sources of biases such as underreporting, overreporting, or inaccurate reporting.26,27 Pertinent clinical information (eg, new medications, new dental fillings/implants) that could potentially misrepresent the actual relationship between vaccination and development of AEs also was not available in the VAERS database. A cohort study with long-term follow-up or a large-scale case-control study would be useful in evaluating such associations.
Conclusion
Lichen planus and LDE can occur, albeit rarely, after vaccination, especially following hepatitis B vaccination. When middle-aged adults present to the clinic with LP or LDE, it is important to inquire about recent vaccination history in addition to a detailed medication history.






