The Surviving Sepsis Campaign: Where have we been and where are we going?
ABSTRACTThe Surviving Sepsis Campaign develops and promotes evidence-based guidelines and performance-improvement practices aimed at reducing deaths from sepsis worldwide. The most recent guidelines, published in 2013, provide detailed management strategies for acute care, fluid resuscitation, and vasopressor use. In addition, the campaign has developed simple, short protocols for what to do within 3 and 6 hours of recognition of sepsis. These protocols are associated with reduced mortality rates.
KEY POINTS
- Ideally, intravenous antibiotic therapy should start within the first hour after sepsis is recognized; performance improvement protocols set a target of within 3 hours.
- A specific source of infection that requires source control measures should be sought, diagnosed or excluded, and if located, treated as rapidly as possible.
- Crystalloids should be used for initial fluid resuscitation. Adding an albumin-based solution is suggested for patients who require substantial amounts of crystalloids.
- Vasopressors are indicated for those who remain hypotensive despite fluid resuscitation. Norepinephrine should be used initially, and if the target mean arterial pressure cannot be achieved, then epinephrine or low-dose vasopressin is added.
- Corticosteroids should be considered only for patients who remain unstable despite adequate fluid resuscitation and vasopressor therapy.
WHAT DRIVES HYPOTENSION IN SEPTIC SHOCK?
In septic shock, mechanisms that can lower the blood pressure include capillary leakage (loss of intravascular volume), decreased arteriolar resistance, decreased cardiac contractility, increased ventricular compliance, and increased venous capacitance (loss of intra-arterial volume).
Capillary leakage ranges from moderate to severe, and it is difficult to know the severity early on during resuscitation. The extent of capillary leakage is often apparent only after 24 hours of fluid resuscitation, when the large amount of fluid needed to maintain intravascular volume produces significant tissue edema. Within the first 24 hours of resuscitation of a patient with septic shock or in the presence of ongoing inflammation, one cannot use intake and output to judge the adequacy of fluid resuscitation.
Reduced arteriolar resistance may be an advantage in the nonhypotensive severely septic patient, compensating for the decreased ejection fraction, but it becomes problematic in the presence of hypotension. In addition, venodilation increases venous capacitance, producing a “sink” for blood and inadequate return of blood volume to the heart.
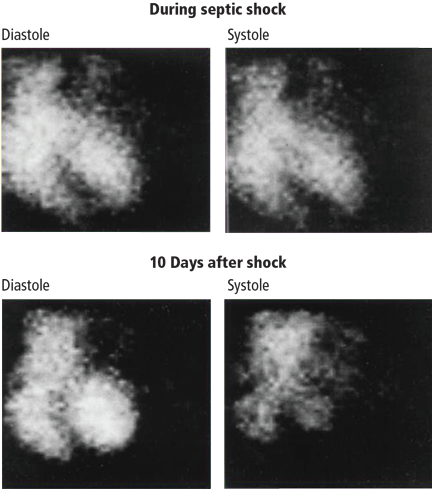
Decreased contractility of the left and right ventricles leads to compensatory sinus tachycardia.12 Reduced heart contractility can be seen by radionuclide angiography: little difference in chamber size is apparent in systole (immediately before contraction) vs diastole (immediately after contraction) (Figure 1).
NOREPINEPHRINE IS THE FIRST-CHOICE VASOPRESSOR
If a patient remains hypotensive after replacement of intravascular volume, the hypotension is due to a combination of vasodilation and reduced contractility, and a combined inotrope-vasopressor is an appropriate drug to raise blood pressure. Therefore, the drug of first choice for raising blood pressure should be a combined inotrope-vasopressor.
There are three combined inotrope-vasopressors: dopamine, norepinephrine, and epinephrine. Head-to-head comparisons of norepinephrine and dopamine have supported a survival advantage with norepinephrine in patients with shock, including septic shock.13 A meta-analysis of six randomized trials totaling 2,768 patients also supports norepinephrine over dopamine in septic shock. Dopamine has been associated with a higher incidence of tachyarrhythmic events.14
Recommendations. Norepinephrine is the first choice for vasopressor therapy (grade 1B). If an additional agent is needed to maintain blood pressure, epinephrine should be added to norepinephrine (grade 2B). Alternatively, vasopressin (0.03 U/minute) can be added to norepinephrine to raise mean arterial pressure to target or to decrease the norepinephrine dose (ungraded recommendation).
Dopamine is not recommended as empiric or additive therapy for septic shock. It may be considered, however, in the presence of septic shock with sinus bradycardia.
Phenylephrine for special cases
Phenylephrine is a pure vasopressor: it decreases stroke volume and is particularly disadvantageous in patients with low cardiac output.
Recommendation. Phenylephrine is not recommended as empiric or additive therapy in the treatment of septic shock, with these exceptions (grade 1C):
- In unusual cases in which norepinephrine is associated with serious tachyarrhythmia, phenylephrine would be the least likely vasopressor to exacerbate arrhythmia
- If cardiac output is known to be high and blood pressure is persistently low
- If it is used as salvage therapy when combined inotrope-vasopressor drugs and low-dose vasopressin have failed to achieve the mean arterial pressure target.
RESUSCITATION OF SEPSIS-INDUCED TISSUE HYPOPERFUSION
A more severe form of sepsis-induced tissue hypoperfusion occurs in patients with severe sepsis, who require vasopressors after fluid challenge or have a lactate level of at least 4 mmol/L (36 mg/dL). Initial resuscitation is of utmost importance in these patients and often is done in the emergency department or regular hospital unit. These patients are targeted for “quantitative resuscitation,” ie, a protocol of fluid therapy and vasoactive agent support to achieve predefined end points.
Rivers et al15 published a landmark study of “early goal-directed therapy” targeting the early management of sepsis-induced tissue hypoperfusion (vasopressor requirement after fluid resuscitation or lactate > 4 mmol/L) and reported significant improvement in the survival rate when resuscitation was targeted to a superior vena cava oxygen saturation of 70%. Both control-group and active-treatment-group patients had central venous pressure targets of 8 mm Hg or greater. The Surviving Sepsis Campaign adopted these targets as recommendations in the original 2004 guidelines and continued through the 2013 guidelines, although the campaign’s sepsis management “bundles” that had originally included specific targets for central venous pressure and central venous oxygen saturation as above were changed in the 2013 guidelines to only measuring these variables (see discussion below).
Jones et al16 analyzed studies that involved early (within 24 hours of presentation) vs late (after 24 hours or unknown) quantitative resuscitation for sepsis-induced tissue hypoperfusion and found a significant reduction in the rate of death with early resuscitation but no difference with late resuscitation compared with standard therapy.
ALTERNATIVES TO MEASURING PRESSURE TO PREDICT RESPONSE TO FLUID
The campaign recognizes the limitation of pressure measurements to predict the response to fluid resuscitation. Some clinicians have objected to the guidelines, arguing that new bedside technology provides better information than central venous pressure or superior vena cava oxygen saturation.
It is useful to recall the Starling principle, which is based on the behavior of isolated myocardial fibrils that are put under the strain of graduated weights and then are stimulated to contract, modeling the contractility of the heart. The more the fibril is stretched, the more intense the contraction. Increased contractility explains why fluid resuscitation increases cardiac output; it is not simply a matter of increasing fluid volume in the veins. Increased volume in the left ventricle increases stretch, causing more intense contractility and higher stroke-volume cardiac output.
The guidelines are based on pressure measurements, but volume is the important measure that drives contractility. For this reason, the 2013 guidelines encourage the use of alternative measures if a hospital has the capability to assess and use them. These alternative measures include changes in pulse pressure, systolic pressure, and stroke volume during the respiratory cycle or with fluid bolus. The greater the variation in these measures, the more likely the patient will respond to additional fluid therapy.17 Normal values:
- Pulse pressure variation: < 13%
- Systolic pressure variation: < 10 mm Hg
- Stroke volume variation: < 10%.
The problem with the more sophisticated technologies is that they tend to be available only in academic centers and not at hospitals doing the critical early resuscitation of septic shock.
The serum lactate level
Measuring serum lactate levels is an alternative method for monitoring resuscitation of early septic shock. This method is widely available even with point-of-care testing. If the lactate level is elevated, quantitative resuscitation, fluids, inotropes, and oxygen delivery can be targeted to lactate clearance.
Recommendation. In patients in whom elevated lactate levels are used as a marker of tissue hypoperfusion, resuscitation should be targeted to normalize lactate as rapidly as possible (grade 2C).






