The role of surgery for locally advanced non–small cell lung cancer
ABSTRACTAccurate clinical staging of patients with locally advanced non–small cell lung cancer (NSCLC) is critical in identifying surgical candidates, who are typically patients with stage N2 disease. Preoperative staging by 18F-fluorodeoxyglucose–positron emission tomography can alter staging and therefore influence the selection of therapy. The staging evaluation should include an assessment of the mediastinal lymph nodes; mediastinal lymph node involvement is a negative prognostic indicator. When the treatment plan potentially includes surgery, multidisciplinary evaluation including physiologic evaluation is essential, as surgery is aggressive with potential morbidity. Multimodality therapy offers the best chance for improved progression-free and overall survival. Patients with potentially resectable NSCLC who are downstaged following induction chemotherapy have a superior prognosis compared with those whose stage is unaltered.
RESULTS OBTAINED WITH MULTIMODALITY THERAPY
Southwest Oncology Group 8805
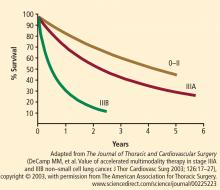
The Southwest Oncology Group (SWOG) study 8805 used a trimodality approach in patients with bulky stage III NSCLC: induction chemoradiation with concurrent cisplatin, etoposide, and radiotherapy (45 Gy) followed by surgical resection.4 The 3-year survival rate with this treatment strategy was 26%. Patients in this trial who were downstaged following induction therapy so that they had node-negative disease at the time of surgery had a superior prognosis, with a 3-year survival rate of 41%. Therefore, a subset of patients with stage III NSCLC stands to benefit from surgery, but identifying this group prior to surgery may not be possible.
Trial of accelerated multimodality therapy
An accelerated multimodality induction regimen given over 12 days was tested in 105 patients with stage IIIa (n = 78) and stage IIIb (n = 27) NSCLC, 97% of whom had mediastinal involvement.5 Seven patients had T4 disease. The induction regimen consisted of a 12-day course of concurrent cisplatin, paclitaxel, and radiotherapy. A 4-day continuous infusion of cisplatin (20 mg/m2/day) and a 24-hour continuous infusion of paclitaxel (175 mg/m2) were administered on day 1. Concurrent accelerated fractionated radiotherapy consisted of twice-daily fractions of 1.5 Gy.
All patients completed induction therapy. Of the 105 patients, 98 were candidates for surgical treatment and 83 underwent curative resections (lobectomy, n = 42; pneumonectomy, n = 36; and bilobectomy, n = 5).
Surgical mortality was 7% and morbidity was 31% (supraventricular arrhythmia, 18%; recurrent laryngeal nerve palsy, 6%; pneumonia or adult respiratory distress syndrome, 3%; bronchopleural fistula, 3%; wound infection, 2%; reoperation for bleeding, 1%).
Profiles of patients with favorable and unfavorable prognoses were developed. A younger patient with adenocarcinoma whose disease was downstaged with induction therapy had a favorable prognosis, whereas an older patient with squamous carcinoma that did not respond to treatment and continued in pathologic stage IIIb had an unfavorable prognosis.
SURVIVAL DATA FAVOR SURGERY
An accurate head-to-head comparison of chemoradiation with or without surgery in patients with resectable NSCLC is difficult because patients selected for surgery must meet performance status criteria, whereas an evaluation of performance status is not mandated for patients treated with definitive chemoradiation alone. The quality of postoperative care and the management of postoperative complications also differ from institution to institution.
A controlled trial in which patients with stage IIIa NSCLC were randomized to chemoradiation with or without surgical resection was performed by Albain et al.6 The induction regimen consisted of 2 cycles of cisplatin and etoposide plus radiotherapy (45 Gy). At 5 years, overall survival was 27% in patients who underwent resection and 20% in those who continued radiotherapy without resection, a difference that did not achieve statistical significance. Progression-free survival was superior in the group assigned to surgery compared with those not undergoing resection (median: 12.8 months vs 10.5 months).
A Surveillance Epidemiology and End Results registry of more than 48,000 patients with stage III NSCLC revealed significantly better overall survival in those who received neoadjuvant radiotherapy plus surgery compared with radiation therapy alone, postoperative radiation therapy, and surgery alone.7
CLEVELAND CLINIC EXPERIENCE WITH ACCELERATED PROTOCOL
At Cleveland Clinic, the current protocol for stage IIIa and IIIb NSCLC is an accelerated multimodality regimen consisting of paclitaxel, 50 mg/m2 twice weekly for 3 weeks; carboplatin (target area under the concentration vs time curve dosing) twice weekly for 3 weeks; and daily erlotinib (phase 1 dose escalation protocol) with concurrent radiotherapy, 1.5 Gy twice daily, as induction therapy, followed by a preoperative evaluation and surgery if local control is achieved with induction treatment.
This protocol has been used in 30 patients with stage IIIa disease (median age: 61 years) with no operative mortality (62% lobectomy, 38% pneumonectomy) and a median length of stay of 6.2 days. Forty percent of patients had their disease downstaged following induction therapy. Three-year survival is approximately 60% and, at 5 years, survival is still 55%.
CONCLUSION
Multimodality therapy for NSCLC is effective and achieves favorable survival. Pathologic downstaging is an important predictor for survival but patients with residual N2 disease still have meaningful survival with resection.
A team approach to evaluation and treatment among medical oncology, radiation oncology, pulmonary medicine, and thoracic surgery is critical to successful outcome.