Off spells and dyskinesias: Pharmacologic management of motor complications
ABSTRACTThere are two major causes of disability in patients with Parkinson disease: motor fluctuations that occur when a dose of levodopa becomes ineffective, leading to a “wearing off,” and hyperkinetic movements (dyskinesias) caused by excessive levels of dopamine. The utility of continuous levodopa treatment is therefore limited by motor complications. Pharmacologic options to treat wearing off include adding (or increasing the dosage of) levodopa, adding (or increasing the dosage of) a dopamine agonist, or adjunctive treatment with a monoamine oxidase inhibitor or catechol-O-methyltransferase inhibitor. Dyskinesias will respond to a reduction in levodopa dosage at the expense of worsening parkinsonism and an increase in the number of “off” episodes. Continuous dopamine stimulation may overcome the pulsatile stimulation of postsynaptic dopamine receptors produced by standard oral formulations of levodopa that lead to motor complications.
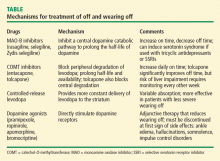
TREATMENT OF OFF AND WEARING OFF
Food and tolerance
The patient should not take levodopa with protein-containing meals, particularly if his or her PD is at an advanced stage. If excessive nausea, vomiting, or lightheadedness prevents the patient from taking an adequate dose, adding carbidopa (up to 75 mg) to the regimen will be helpful.
MAO-B inhibitors
By inhibiting one of the central dopamine catabolic pathways, MAO-B inhibitors (selegiline, rasagiline, and Zydis selegiline) prolong the half-life of dopamine in the brain and increase on time.
Improvement in off time with rasagiline is comparable to that seen with the COMT inhibitor entacapone. In an 18-week, double-blind trial of 687 patients randomized to receive once-daily rasagiline, entacapone, or placebo as an adjunct to levodopa, both rasagiline and entacapone reduced off time by 1.2 hours compared with placebo.3
In a 26-week placebo-controlled study, rasagiline decreased off time by 29% when added to levodopa in patients with PD and motor fluctuations, compared with a 15% reduction in the placebo group.4 This study confirmed the benefit of adding rasagiline to the regimens of patients who were already optimally treated with levodopa, dopamine agonists, amantadine, anticholinergics, and entacapone before enrolling in the study.
An orally disintegrating selegiline (Zydis selegiline) tablet is particularly useful for patients who have difficulty swallowing. The bioavailability of Zydis selegiline is 80% compared with 10% for selegiline, resulting in faster absorption. Pregastric absorption of Zydis selegiline avoids extensive first-pass metabolism in the liver and, therefore, the concentration of amphetamine-like metabolites is much lower.
In a 3-month, placebo-controlled study of patients with PD who were experiencing levodopa-related motor fluctuations, Zydis selegiline was associated with a 2.2-hour reduction in the total number of off hours compared with 0.6 hours in the placebo group, and dyskinesia-free on hours increased by 1.8 hours.5
The use of MAO-B inhibitors with tricyclic antidepressants or selective serotonin reuptake inhibitors has been reported to induce the serotonin syndrome by activation of 5HT1a and 5HT2 receptors. Serotonin syndrome is a potentially life-threatening accumulation of serotonin that can cause encephalopathy, severe rigidity of the legs, dysautonomia (diarrhea, mydriasis, and excessive lacrimation), myoclonus, hyperreflexia, and seizures.
COMT inhibitors
Catechol-O-methyltransferase inhibitors (entacapone and tolcapone) block peripheral degradation of levodopa. Tolcapone also blocks central degradation of levodopa and dopamine. These mechanisms increase central levodopa and dopamine levels and prolong levodopa half-life and bioavailability. Tolcapone has more powerful COMT inhibition than entacapone because tolcapone crosses the blood-brain barrier and inhibits the peripheral and central pathways of levodopa degradation. Use of COMT inhibitors can increase daily on time, but diarrhea is a common side effect and leads to withdrawal of these agents in about 3% of patients.
Tolcapone-treated patients show significant improvement in off time with improvement in motor fluctuations.6 Because tolcapone causes rare instances of fulminant hepatitis, liver function needs to be monitored every other week. For this reason, tolcapone should be reserved for patients in whom other treatments, including entacapone, have failed.
Controlled-release levodopa
Controlled-release levodopa was developed to provide more constant delivery of levodopa to the striatum. The benefit of controlled-release levodopa is only mild, however, as absorption of this formulation is variable. In advanced PD cases, the effects of controlled-release levodopa are more unpredictable than those with standard levodopa. Controlled-release levodopa is effective in patients with less severe wearing off, but it is not as effective in patients with a less predictable pattern of fluctuations.
Dopamine agonists
Dopamine agonists (pramipexole, ropinirole, apomorphine, and bromocriptine) have shown beneficial effects as adjunctive therapy to reduce wearing off. Side effects of dopamine agonists include ankle edema, hallucinations, somnolence, and impulse control disorders. These side effects should be discussed with patients before instituting therapy, and therapy should be discontinued if any of them occur.
In patients with advanced PD, pramipexole was shown to improve motor function during on and off periods, decrease the total off time, and decrease the severity of off time. Further, a larger reduction in the dosage of levodopa was possible in the pramipexole-treated patients than in the placebo-treated patients.7
In a comparison of pramipexole with levodopa on the end point of motor complications of PD in 300 patients, the incidences of wearing off and dyskinesia were significantly lower in the patients randomized to pramipexole with follow-up over 4 years.8 Only 25% of patients initially treated with pramipexole exhibited dyskinesia compared with 54% of patients initially treated with levodopa. Forty-seven percent of patients in the pramipexole group experienced wearing off compared with 63% initially treated with levodopa. Pramipexole is available as tablets ranging from 0.125 mg to 1.5 mg in size. It is given in three divided daily doses with gradual increments of 0.25 mg three times a day every week. Pramipexole is now also available in an extended-release formulation for once-a-day dosing in tablets ranging in size from 0.375 mg to 4.5 mg.
Ropinirole adds clinical benefit in PD patients with motor fluctuations and also permits a reduction in the dosage of levodopa.9
In one study, ropinirole monotherapy was compared with levodopa therapy in 268 patients with early PD. By the end of the 5-year study, 45% of the levodopa patients experienced dyskinesias versus 20% of the ropinirole patients.10
Ropinirole is available as tablets ranging in size from 0.25 mg to 5 mg. It is now also available in an extended-release (XL) formulation, with tablet sizes ranging from 2 mg to 12 mg. Ropinirole XL is taken once a day.
Bromocriptine is an old ergot-derived dopamine agonist that has also been studied for monotherapy and add-on treatment in PD. Due to the potential risks of pulmonary, retroperitoneal, and heart valve fibrosis, bromocriptine is not commonly used.
Apomorphine was approved by the US Food and Drug Administration in 2004 as an acute, intermittent, subcutaneous injection for the treatment of hypomobility off episodes (end-of-dose wearing off and unpredictable on-off episodes) associated with advanced PD. Apomorphine has been shown to be beneficial in patients with unpredictable off periods.11 Its onset of action is 10 to 15 minutes, and the effects of each dose last for 60 to 90 minutes. The best tolerated dose is 4 mg to 10 mg. Apomorphine appears to be most useful as as rescue medication in the refractory off periods with severe bradykinesia and unpredictable off periods.