PML and rheumatology: The contribution of disease and drugs
ABSTRACTProgressive multifocal leukoencephalopathy (PML), a rare, typically fatal, opportunistic infection caused by the JC virus, is becoming relevant to physicians in multiple specialties, including those who prescribe biologic agents for the treatment of autoimmune disorders. Reports of PML have led to US Food and Drug Administration alerts and warning letters regarding four immunosuppressive agents in recent years (natalizumab, rituximab, efalizumab, and mycophenolate mofetil). Consequently, informed clinical decision-making requires understanding the risk of PML associated with these therapies. An estimate of the relative frequency of PML associated with specific rheumatic conditions has been generated. Systemic lupus erythematosus appears to be associated with susceptibility to PML that cannot be fully explained by the intensity of immunosuppressive therapy. Further, the use of rituximab in patients with rheumatic disease has raised concerns. However, definitive attribution of cause is precluded by the limitations of the currently available data. All patients with rheumatic disease, regardless of the intensity of their current immunosuppressive therapy, should be considered potentially at risk of PML. With an evolving understanding of a greater clinical heterogeneity of PML, advances in diagnostic methods, and significant implications for therapy, PML should be considered in the differential diagnosis of neurologic manifestations of rheumatic diseases.
Efalizumab
Efalizumab is a monoclonal antibody that targets CD11a, the alpha subunit of lymphocyte function–associated antigen 1. Efalizumab blocks binding to intercellular adhesion molecule 1, and thereby blocks T-cell adhesion and migration. CD11a is also expressed on a variety of other leukocytes and lymphocytes such as B cells, monocytes, and natural killer cells.
Efalizumab was approved in 2003 by the FDA for the treatment of moderate to severe plaque psoriasis. It is estimated that 46,000 patients have been treated with efalizumab worldwide since its approval. In 2008, a black box warning was added to the efalizumab prescribing information following the occurrence of serious infections, including pulmonary tuberculosis, necrotizing fasciitis, and invasive fungal infections.8 Subsequently, four cases of PML, three of which were fatal, were reported in psoriasis patients treated with efalizumab. Of note, these were the first cases of PML reported in patients with psoriasis. Of more concern, the affected patients were among a group of approximately 1,100 patients who had been treated with efalizumab for more than 3 years. In February 2009, a public health advisory was issued by the FDA,9 and efalizumab was voluntarily withdrawn by its manufacturer 2 months later.
Belatacept
Belatacept is a recombinant soluble fusion protein of the extracellular domain of human cytotoxic T-lymphocyte antigen-4 with a fragment of a modified Fc domain of immunoglobulin G1. Recently approved by the FDA for prophylaxis of renal transplant rejection, it is a second-generation, higher-avidity version of abatacept. Abatacept is licensed for the treatment of rheumatoid arthritis and is under investigation for the treatment of vasculitis and SLE. Belatacept differs from abatacept by only two amino acids.
Two cases of PML have been reported in association with belatacept, one in a patient following renal transplantation and the other in a patient following liver transplantation. Both patients had been treated with other standard immunosuppressive therapies for prophylaxis of organ transplant rejection, including mycophenolate mofetil.
Mycophenolate mofetil
Mycophenolate mofetil is the prodrug of mycophenolic acid. Both have been the subjects of FDA alerts regarding PML, based on a January 2008 report of 10 definite and 7 possible cases of PML occurring with mycophenolate mofetil. The patients affected included four with SLE, none of whom underwent a renal transplant.10
In a retrospective cohort study of 32,757 renal transplant patients, Neff et al11 found 14 cases of PML per 100,000 person-years among patients treated with mycophenolate mofetil following kidney transplant compared with none in patients who did not receive mycophenolate mofetil. It is difficult to ascertain risk with mycophenolate mofetil because it is standard therapy among renal transplant patients, leaving few patients in these groups unexposed.
Given the FDA alert with respect to mycophenolate mofetil and PML,10 the frequent use of mycophenolate mofetil in the setting of SLE, and the concerns about possible predisposition to PML among patients with SLE, it will be important to clarify the level of risk in patients with SLE who are treated with mycophenolate mofetil.
AGGREGATE EXPERIENCE: REVIEW OF FEDERAL DATABASE
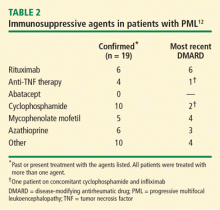
Ten cases of PML were confirmed with cyclophosphamide treatment, and cyclophosphamide was the most recent DMARD prescribed in two of these cases. Five cases were confirmed with mycophenolate mofetil (in four of which it was the most recently prescribed DMARD) and six with azathioprine (in three of which it was the most recently prescribed DMARD).
Risk of PML with DMARD therapy
Rituximab. The confirmation of six cases of PML among rituximab-treated rheumatoid arthritis patients is a source of concern. Nevertheless, PML is a rare adverse event. It occurs in fewer than 1 in 10,000 rituximab-treated patients who have rheumatoid arthritis, among a total of approximately 130,000 such patients. A better understanding of the potential mechanism responsible for the increased risk of developing PML may help in risk prediction and to guide patient selection for this agent.
Anti-TNF therapy. A paucity of confirmed cases in patients treated with anti-TNF therapy argues against a significant risk of PML associated with this therapy, especially considering the estimated 2 to 3 million rheumatoid arthritis patients who are receiving treatment with anti-TNF agents. A note of caution is sounded by a recent case report of PML in a rheumatoid arthritis patient. The patient had been treated with infliximab, with the only background therapy being methotrexate.13 Ongoing vigilance is therefore necessary.
Mycophenolate mofetil. All five confirmed cases of PML in mycophenolate mofetil-treated patients had earlier received treatment with cyclophosphamide. These data indicate no clear signal of excess risk with mycophenolate mofetil above that seen with other nonbiologic immunosuppressive agents, such as cyclophosphamide or azathioprine.
CONCLUSION AND RECOMMENDATIONS
PML has been reported in association with a variety of disease states, although a predisposition in patients with SLE has become apparent. Synthetic and biologic immunosuppressive therapies have also been implicated, but PML may also occur in the setting of minimal iatrogenic immunosuppression.
Until greater clarity can be achieved, all patients with systemic rheumatic diseases should be considered at risk for PML, regardless of the nature or intensity of their immunosuppressive therapy. In this context, differentiating PML from neurologic syndromes related to the underlying rheumatic disease (eg, neuropsychiatric SLE, cerebral vasculitis) is critical, particularly given the markedly different approaches to management.
PML should be considered in patients with unexplained subacute progressive focal and diffuse neurologic deficits, especially if their clinical or radiologic status worsens in the face of increased intensity of immunosuppressive therapy. Spinal cord or optic nerve involvement argues against PML. A normal magnetic resonance image (MRI) has a high negative predictive value, and frank infarction is not a feature of PML. In classic PML, contrast enhancement is typically absent and routine cerebrospinal fluid (CSF) analysis is typically normal. However, contrast enhancement and edema on MRI, lymphocytic CSF pleocytosis, and elevated CSF protein may be seen in the more recently described “inflammatory PML,” in which case the distinction from cerebral vasculitis or neuropsychiatric SLE may be more difficult. Angiography appears normal in patients with PML.
The diagnostic test of choice is a polymerase chain reaction (PCR) assay for JCV in CSF. If the PCR is repeatedly negative, then a brain biopsy should be considered, especially in the setting of progressive neurologic decline in patients receiving immunosuppressive therapy.