Multiple sclerosis, natalizumab, and PML: Helping patients decide
ABSTRACTThe treatment benefits of natalizumab in patients with multiple sclerosis (MS) appear to exceed those of other disease-modifying drugs, but progressive multifocal leukoencephalopathy (PML) has been identified as a risk in patients receiving natalizumab. As of August 2011, a total of 150 cases of natalizumab-associated PML had been reported worldwide. The overall risk is estimated at approximately 1.66 in 1,000 patients. Independent risk factors for natalizumab-associated PML are number of infusions beyond 36 and prior use of immunosuppressive drugs. Classifying JC virus antibody status appears to be useful in treatment decision-making for individual MS patients. Patient tolerance for risk plays an important role in the selection of therapy, and the treating physician’s perception and tolerance of risk may differ markedly from the patient’s. Physicians can help patients make individual informed decisions regarding the use of natalizumab, given the known risk of PML.
Risk related to duration of therapy
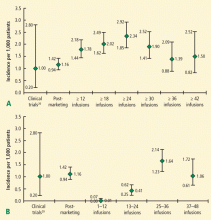
Of the first 35 cases of natalizumab-associated PML, 10 cases (29%) were fatal. Among surviving patients, the level of disability was found to be severe in 48%; moderate in 36%; and mild in 16%.23 Improved survival was associated with younger age, less MS-related disability prior to PML, more localized disease on brain MRI at diagnosis, and shorter time from symptom onset to PML diagnosis.23 As of August 4, 2011, there were 150 confirmed cases of natalizumab-associated PML (58 in the United States, 85 in Europe, and 7 from the rest of the world); of these, 29 (19%) have died.22
VARIATION IN PATIENT RISK TOLERANCE
The postmarketing surveillance of natalizumab clearly demonstrates that risk is associated with administration of the drug, but risk tolerance varies considerably among individuals with MS. Some patients elect to use natalizumab despite the risk of PML, even when they have relatively mild MS. Other patients decline use of natalizumab even when their MS is severe and has responded poorly to other disease-modifying drugs.
In most cases, based on my experience, patients accept the risk of natalizumab-associated PML if MS disease is their primary consideration. Another major factor is the patient’s prior experiences with disease-modifying drugs; patients who have experienced breakthrough disease activity despite treatment with first-line drugs commonly opt for natalizumab regardless of the risk of PML.
Interestingly, the treating neurologist’s perception of MS severity and risk of PML may differ from the patient’s perception. In a study of 69 natalizumab-treated MS patients and 66 neurologists, Heesen et al found that patients had a significantly worse perception of their disease and were more willing to assume treatment risks and continue natalizumab therapy than their neurologists were.24 About one-half of the neurologists said that they would discontinue natalizumab at a risk level of 1 in 5,000 or lower, whereas only 17% of the patients would stop at this risk level. This finding has significant implications for clinical practice and implies that the neurologist should discuss concerns about MS and risk of treatment with the patient in order to tailor the decision to the patient’s concerns.
Interest in identifying biomarkers to aid in quantifying risk is ongoing. Chen et al found that subclinical reactivation of the JC virus (JCV) occurred frequently in 19 natalizumab-treated MS patients.25 Another study of 24 natalizumab-treated MS patients found no JCV DNA in the blood, although JCV DNA was found in the urine in 25% of patients.26 A large survey of blood and urine from natalizumab-treated MS patients found low sensitivity and specificity for JCV DNA as a predictor for subsequent PML.27 In this study of more than 1,000 natalizumab-treated patients, JCV DNA was detected in 0.3% of patients’ plasma and in 26% of patients’ urine, but PML did not develop in any patient who was JCV-positive. Among five natalizumab-treated patients who developed PML, JCV DNA was detected in none before the advent of symptoms. The presence of JCV DNA in bodily fluids is important for the diagnosis of PML, but it currently holds no predictive clinical value. At present, measuring JCV DNA in blood, cells, or urine as a predictive biomarker for natalizumab-associated PML does not appear to be clinically useful.
Stratifying risk by measuring JCV serology, however, does appear to be a useful strategy. Investigators using a two-step assay to detect and quantify JCV antibodies found 53.6% of MS patients to be seropositive, with a false-negative rate of 2.5%. Of most interest, all 17 natalizumab-associated PML patients who had available blood samples taken an average of 2 years before onset of PML tested positive for JCV antibodies.28 Although studies are ongoing, classification according to JCV-antibody status may be helpful in advising patients. Patients who are JCV-antibody seronegative (about one-half of patients) appear to be at extraordinarily low risk for PML. In these patients, use of natalizumab could be liberalized and continued as long as the JCV-antibody status remains negative. In patients who are seropositive for JCV antibodies, caution is recommended, particularly for patients who had prior immunosuppressive drug therapy and for patients who have received treatment for more than 24 months. Even in JCV-antibody–seropositive patients, use of natalizumab may be advisable depending on disease severity, available options, and the patient’s risk tolerance. JCV-antibody testing is a rare example of a clinically useful biomarker that can guide specific treatment decisions in the field of neurology.
CURRENT PRACTICE: A PERSONAL MANAGEMENT ALGORITHM
Based on current evidence, the following opinion on the use of natalizumab for the treatment of MS is offered as a supplement to approved prescribing information. The neurologist must individualize the treatment decision for each patient and recognize that these general comments represent a personal opinion. Several factors affect decisions about the use of disease-modifying drugs in MS, and specifically use of natalizumab: How severe is the disease, and what is the prognosis for future disease progression from the neurologist’s perspective? How concerned is the patient about current or future MS symptoms and disability? What is the patient’s tolerance for medication side effects? For risk taking? Has there been prior immunosuppressive therapy? What is the JCV antibody status? What other options are available for disease management?
These issues require discussion among the neurologist, the patient, and the patient’s family. The neurologist should provide input on disease status, an opinion about prognosis, and a description of appropriate options for disease management. Many patients also want a global recommendation (ie, “Tell me what you think I should do”). The neurologist must tailor that global recommendation to the patient’s perceptions of his or her MS, its treatment, and preferences regarding treatment options.
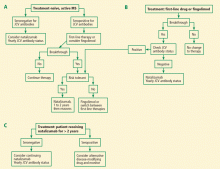
For patients who are already receiving treatment with a first-line drug and whose disease is well controlled, I make no changes in treatment until a breakthrough occurs, at which time I recommend switching to natalizumab with JCV antibody status assessed yearly (Figure 2B). If a patient has been taking natalizumab for more than 2 years and is seronegative, I advise continuing natalizumab (Figure 2C). If seropositive after 2 or more years of natalizumab therapy, I recommend switching to fingolimod and monitoring for disease reactivation.