Treatment of chronic inflammatory diseases with implantable medical devices*
ABSTRACTImplantable medical devices are finding increasing use in the treatment of diseases traditionally targeted with drugs. It is well established that the cholinergic anti-inflammatory pathway serves as a physiological regulator of inflammatory responses, but stimulation of this pathway therapeutically by electrical stimulation of the vagus nerve can also diminish excessive or dysregulated states of inflammation. Recent data from a wide variety of animal models, as well as evidence of reduced vagal tone in rheumatoid arthritis and other inflammatory diseases, support the rationale for, and feasibility of, developing implantable vagal nerve stimulation devices to treat chronic inflammation in humans.
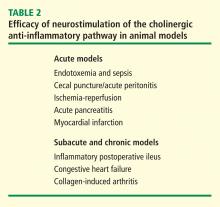
NEUROSTIMULATION OF THE CAP IN ANIMAL MODELS OF DISEASE
In a canine model of CHF induced by rapid ventricular pacing, inflammation and ventricular remodeling with fibrosis are typically accompanied by marked increases in serum C-reactive protein (CRP) levels. In addition to improving the physiological manifestations of CHF, VNS resulted in 60% to 80% reductions in CRP for up to 8 weeks.17 In another canine CHF model induced by repetitive microembolization, which is similarly associated with systemic and myocardial inflammation, VNS markedly reduced circulating levels of interleukin 6 and TNF for up to 12 weeks.18 Importantly, both these studies show that rapid tachyphylaxis does not appear to occur with NCAP over periods of time that are relatively chronic by the typical standards of animal models.
VAGAL NERVE STIMULATION FOR EPILEPSY AND DEPRESSION: EXPERIENCE IN HUMANS
VNS delivered using a surgically implanted cuffed cervical vagus nerve lead and pacemaker-style pulse generator device has been approved for the treatment of refractory epilepsy in the United States since the mid-1990s and has more recently been approved for treatment of depression. Over 50,000 patients have been implanted with these devices world wide since that time. The safety profile of both surgical implantation and VNS delivery in this setting is well established.19 The major tolerability problem is laryngeal and pharyngeal symptoms, such as hoarseness and dysphonia, which are present almost solely during periods of active device stimulation. The frequency and severity of these treatments decreases after receiving treatment for an extended time.20 With growing experience in VNS delivery over the first 5 years of use, it also became apparent that reducing the active stimulation duty cycle from 40% to 10%, and keeping stimulation currents at ≤ 1.5 mA results in a marked reduction in these symptoms.21 Of note, the stimulation currents necessary to evoke NCAP in animals are well below the 1.5 mA level, and as above, NCAP is effective even with very brief, once-daily periods of stimulation (ie, a duty cycle of 0.07% if given for 1 min each day). Thus it is likely that the laryngeal and pharyngeal adverse event profile of VNS will not be problematic in the setting of NCAP delivery for inflammation.
A POTENTIAL ROLE FOR THERAPEUTIC NCAP USING IMPLANTABLE DEVICES IN HUMAN INFLAMMATORY DISEASES
Autonomic nervous system activity can be measured indirectly by recording cardiac R-R interval variability and subjecting the data to power spectral analysis. Such heart rate variability (HRV) measurements are influenced by the levels of vagus nerve activity and by balance in cardiac sympathetic–parasympathetic tone. Reduced HRV is indicative of decreased vagal tone, and reductions in HRV have a strong inverse correlation with CRP levels, progression of atherosclerosis, and risk of sudden death.22,23 HRV is also reduced relative to normal subjects in patients with RA, systemic lupus erythematosus, and Sjögren syndrome, and the extent of reduction in HRV within the patient groups correlates with disease severity.24–26 Although these associations are only correlative and do not provide firm evidence of causality, they do provide additional epidemiological support for the hypothesis that driving increased vagal activity using implantable devices may have a favorable effect on inflammatory disease.
Implantable neurostimulation devices have not yet been tested in human patients with RA. However, preliminary evidence from a small study carried out in normal volunteers demonstrated that the CAP reflex can be elicited by brief mechanical stimulation of the afferent auricular branch of the vagus nerve, as shown by reduction of in vitro LPS-inducible cytokine production (T van der Poll, personal communication). Clinical testing of NCAP using implantable VNS devices will begin in the near future. The devices to be used for these initial studies will be very similar in design to those currently in use for epilepsy treatment. However, prototype versions of the device which will be used in follow-on studies are miniaturized to the point where they will be directly implantable on the vagus nerve, without the need for a pulse generator unit on the chest and will use a small self-contained battery system which can be recharged using transcutaneous radiofrequency induction. Given the long lifespan, relatively low cost, and potential for increased safety over currently available treatments, NCAP delivered using an implantable device holds great promise as a novel potential therapeutic approach for patients with RA and other inflammatory diseases.