New fecal occult blood tests may improve adherence and mortality rates
ABSTRACTSeveral new fecal occult blood tests have advantages over older ones when used for colorectal cancer screening. Fecal immunochemical tests can detect antibodies to human globin in the stool and can be used without the dietary restrictions needed with traditional guaiac tests. Although colonoscopy is often considered the gold standard, we hope that these new tests will allow more people to be screened and more cases of colorectal cancer to be detected early.
KEY POINTS
- Hemoccult Sensa and several fecal immunochemical tests are more sensitive than Hemoccult and Hemoccult II for detecting colorectal cancer and advanced adenomas, with similar specificity.
- In most screening studies, fecal immunochemical tests have been more sensitive than guaiac-based tests. In addition, rates of adherence were higher, likely because dietary and medication restrictions are not needed and fewer stool samples are required.
- Better compliance should improve participation in colorectal cancer screening and reduce colorectal cancer mortality rates.
New fecal occult blood tests hold promise for improving our detection of colorectal cancer and for lowering mortality rates. This is good news, because despite the proven benefit of being screened for colorectal cancer,1 only an average of 62% of eligible adults are screened,2 and colorectal cancer remains the third leading cause of cancer deaths in the United States.
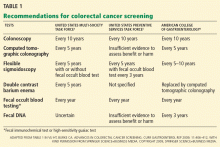
Colonoscopy is often considered the gold-standard screening test for colorectal cancer. However, many patients do not undergo screening colonoscopy because it is invasive and uncomfortable, bowel preparation poses a challenge, the procedure has risks, and it is costly. Members of minority groups, people of lower socioeconomic status, and those who lack health insurance are less likely to undergo screening.
While fecal occult blood tests are cheaper and less invasive than colonoscopy, they do not allow us to prevent colorectal cancer by removing adenomatous polyps. Still, randomized controlled trials have proven that fecal occult blood testing is associated with a decrease in the rate of death from colorectal cancer,3 and it has been shown to be cost-effective.
The challenge is that all guaiac-based tests (gFOBTs), even the newest one, require strict dietary and medication restrictions to be accurate, and the difficulty of collecting stool specimens often results in either false-positive results or failure to complete the test.
The newer tests—one guaiac-based test and several fecal immunochemical tests (FITs)—are more sensitive, and the FITs are more convenient for patients to use than the older guaiac-based tests, advantages that, we hope, will increase the rates of compliance with testing.
GUAIAC-BASED TESTS
Guaiac tests detect the peroxidase activity of hemoglobin. If hemoglobin is present in stool, it catalyzes the oxidation of the active compound in guaiac paper when a hydrogen peroxide developer is added. The resultant conjugated compound is blue.
The lower-sensitivity guaiac tests are commercially available as Hemoccult and Hemoccult II, and the higher-sensitivity guaiac test is Hemoccult Sensa, which has a lower threshold for detecting peroxidase. All are made by Beckman Coulter, Fullerton, CA.
Disadvantages of guaiac tests. Guaiac tests can give false-positive results by detecting pseudoperoxidases in fruits, vegetables, and nonhuman blood. In addition, they can give false-negative results in people who take excessive amounts of vitamin C, which can inhibit peroxidase activity. Therefore, patients need to follow certain dietary restrictions before testing.
Another disadvantage of guaiac tests is that they cannot differentiate between blood lost from the stomach, small bowel, or colon.
Moreover, the interpretation of guaiac tests is subject to observer variation.
Since testing involves dietary restrictions and obtaining two specimens each from three separate stools, patient compliance is poor.
Patient instructions. Patients undergoing guaiac-based fecal occult blood testing should not take nonsteroidal anti-inflammatory drugs (eg, > one adult aspirin per day) for 7 days before and during the stool collection period to avoid causing gastrointestinal bleeding. They should also not eat red meat or take vitamin C in excess of 250 mg/day for 3 days before testing and throughout the test period.
Two specimens are collected from three different stools with a wooden stick and are smeared onto the stool test card, which is then closed and returned to the physician’s office. The specimens must be collected before the stool comes into contact with the toilet water.
Efficacy of guaiac testing
Randomized, controlled trials of guaiac-based fecal occult blood testing have shown a decrease in colorectal cancer incidence.8–11
A Cochrane review12 involved more than 320,000 people in Denmark, Sweden, the United States, and the United Kingdom who underwent testing every year or every 2 years with Hemoccult or Hemoccult II. The primary analysis was by intention to treat, and it showed that participants allocated to screening had a 16% reduction in the relative risk of death from colorectal cancer, or 0.1 to 0.2 fewer colorectal cancer deaths per 1,000 patient-years. The secondary analysis was adjusted for whether the participants actually were screened; the risk reduction in death from colorectal cancer was 25% in participants who attended at least one round of screening.