Use of deep brain stimulation in treatment-resistant depression
ABSTRACT
Deep brain stimulation has emerged as an experimental treatment option for the sizeable proportion of patients with major depression that is refractory to multiple medications and psychotherapy. Chronic stimulation of the ventral internal capsule/ventral striatum has been shown to improve function and mood in patients with severe obsessive-compulsive disorder, and has likewise been applied to patients with treatment-resistant depression. Multicenter experience with chronic deep brain stimulation of the ventral capsule/ventral striatum in 17 patients with severe treatment-resistant depression has demonstrated sustained improvements across multiple scales of depression, anxiety, and global function. Further research on deep brain stimulation in larger populations of patients with treatment-refractory depression is under way. While such research should benefit from the recent US Food and Drug Administration approval of deep brain stimulation for severe obsessive-compulsive disorder, it must adhere to strict principles for appropriate patient selection.
MULTICENTER STUDY OF STIMULATION FOR HIGHLY REFRACTORY DEPRESSION
Our team at Cleveland Clinic partnered with colleagues from Brown Medical School and Massachusetts General Hospital to build on these pilot study findings and evaluate DBS of the VC/VS in patients with chronic, severe refractory depression in a multicenter investigation. The results from the first 15 patients in this series were published in early 2009;6 results from an additional 2 patients, for a total sample of 17, are now available and summarized below.
Patients and study design
Patients had at least a 5-year history of chronic or recurrent depression that was refractory to at least five courses of medication, an adequate trial of psychotherapy, and at least one trial of bilateral electroconvulsive therapy. Exclusion criteria included significant substance abuse, severe personality disorder that could potentially affect safety or compliance, and psychotic depression.
Outcome measures included the Hamilton Depression Rating Scale (HDRS), the Montgomery-Asberg Depression Rating Scale (MADRS), and the Global Assessment of Function Scale (GAF). Assessments were performed at baseline, postoperatively, and monthly thereafter. A detailed neuropsychological battery was performed at baseline and again at 6 months.
At the time of electrode implantation, mean patient age was 46.3 years and mean duration of illness was 21.0 years. In their current depressive episode, patients had had an average of 6.1 antidepressant trials and 6.1 trials of augmentation or combination of antidepressant medications. The average number of lifetime electroconvulsive therapy treatments was 30.5; of the 17 patients, 15 had an adequate trial of electroconvulsive therapy in their current depressive episode.
Electrodes were implanted bilaterally in the VC/VS. Following a postoperative recovery phase of 2 to 4 weeks, stimulation parameters were titrated over several days on an outpatient basis. The stimulation parameters were selected on the basis of positive mood benefit and absence of adverse effects. Stimulation was at a frequency of 100 to 130 Hz and an amplitude of 2.5 to 8 V. These stimulation amplitudes are higher than those used for treatment of movement disorders, reflecting the different targets (white matter vs gray matter) for the different conditions.
The two ventral contacts (referred to as contact 0 and contact 1) tend to be the most active, providing the best response. Contact 0 is the most distal contact, at the VS below the level of the anterior commissure. Contact 1 is near the junction of the VS and VC.
The time to battery replacement (due to depletion) ranged from 10 to 18 months.
Outcomes
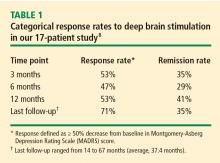
Patients’ mean baseline MADRS score was 34.7, indicating very severe depression. The mean MADRS score improved to 20.6 by 1 month, and declined further to 16.0 at 3 months. This benefit has been maintained to the most recent follow-up (average, 37.4 months; range, 14–67 months). Similar improvements from baseline to most recent follow-up were observed in the HDRS and GAF scores. Additionally, a substantial reduction in suicidality (as measured by mean MADRS suicide subscale score) was observed by 1 month (P = .01) and was maintained through 12 months of follow-up (P < .001).7
Adverse effects
Adverse effects of DBS were observed on occasion and can generally be divided into those related to surgical implantation and those related to stimulation itself.
Effects related to surgical implantation have included infection from lead or battery implantation, adverse cosmetic effects (from placement of the battery-operated neurostimulators into the chest), and repeat surgeries for neurostimulator replacements. A rechargeable battery has recently become available and should enhance tolerability and acceptability by reducing the frequency of replacement surgeries.
Stimulation-induced acute adverse events have included paresthesias, anxiety, mood changes, and autonomic effects. All were reversible with adjustment of stimulation parameters.
DIRECTION AND GUIDANCE FOR FUTURE RESEARCH
A number of recent developments have enhanced the prospects for better understanding of the use of DBS in treatment-refractory depression:
- The recent FDA approval of DBS for severe, refractory OCD should broaden the base of experience with DBS for psychiatric disorders.
- Two large studies of DBS of the VC/VS and the area 25/subgenual cingulate region in depressed patients are currently under way.
- The aforementioned recent development of a rechargeable stimulator battery should improve patient acceptance of DBS therapy.
- Neuroimaging studies using functional magnetic resonance imaging and positron emission tomography may help to elucidate neuroanatomic pathways in depression and other psychiatric disorders.
- Ongoing studies of DBS for Tourette syndrome will broaden the experience base with DBS and perhaps yield insights for depression.
As investigation of DBS for depression moves forward, it must be conducted in keeping with some basic principles for patient selection and fundamental ethical guidelines, especially in light of the troubled early history of psychosurgery. From the individual patient perspective, patients should be selected only if they meet the following criteria:
- Accurate diagnosis. This may seem obvious, yet inaccurate diagnoses in the psychiatric realm are far more widespread than is appreciated but clearly must be avoided when embarking on an intervention as significant as DBS.
- Ability to provide informed consent
- Sufficient severity of illness
- Nonresponse to less-invasive options (ie, reasonable trials of both pharmacotherapy and psychotherapy).
From the ethical and procedural perspective, research of DBS for psychiatric disease must ensure the involvement of expert and dedicated psychiatric neurosurgery teams, led by psychiatrists, as well as full ethical review (by institutional review boards) and method-safety review (in keeping with FDA policy). Additionally, expert centers must be prepared to make the long-term commitment necessary to follow these difficult-to-treat patients. Centers and investigators must also ensure that DBS be used only to alleviate suffering and improve patients’ lives, and never to “augment” normal function or for social or political reasons.9