Recovery of consciousness after severe brain injury: The role of arousal regulation mechanisms and some speculation on the heart-brain interface
ABSTRACT
Recovery of consciousness after severe brain injury involves reconstitution of brain arousal mechanisms and cerebral integrative function. This review discusses several aspects of neuroanatomy and neuropathology relevant to the process of recovery. Particular emphasis is placed on the role of the anterior forebrain and circuit mechanisms linking the frontal lobe, striatum, and central thalamus. The article concludes with some observations on the heart-brain interface and future research directions in the context of recovery from severe brain injury.
WHY WE NEED TO UNDERSTAND MECHANISMS OF RECOVERY AFTER SEVERE BRAIN INJURY
The problem of recovery of consciousness after severe brain injury is one that easily captures the imagination of both the lay public and the professional. Puzzling reports continue to arise of late recovery of speech, language, memory, and other higher cognitive functions in rare patients, yet a scientific framework for the systematic assessment of these phenomena has been lacking.1–3 Some of these cases provide intriguing hints to the possible role of various medications (such as dopaminergic, serotoninergic, and noradrenergic agents) as well as spontaneous changes in brain function arising over time. As discussed below, the varying levels of recovery following coma seen after multifocal traumatic or nontraumatic brain injuries may share some common underlying mechanisms at the “circuit” level. Severe brain injuries producing coma have many causes (see Posner et al4 for a comprehensive review), but careful review reveals an overlap of structural pathologies and functional disturbances isolated to specific cerebral structures across several clinical syndromes grouped under the framework of “disorders of consciousness,” 5 with an emphasis on the role of particular substructures. Perhaps most important is a consideration of the pathologic, anatomic, and pathophysiologic role of the anterior forebrain, particularly the relationships of the brainstem and basal forebrain arousal systems, the central thalamus, and frontostriatal pathways, as reviewed below.
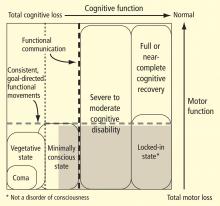
The large gray box in Figure 1 indicates the disquietingly high degree of uncertainty in assessing cognitive level in some patients who lack controllable motor output channels. The locked-in state (bottom right corner of figure) defines patients who retain total preservation of cognitive function but otherwise may appear no different from those in deep coma. Although locked-in state often arises in the context of neurologic injuries that selectively damage motor output pathways distal from their cortical origins or that slowly reduce primary motor neuron function, this syndrome and closely similar conditions arise in patients with complex brain injuries. Such patients likely retain full or nearly normal consciousness but unfortunately are unable to produce consistent goal-directed movements that allow for communication. In principle, such patients could retain significant cognitive capacity near the normal range of cognitive function yet be indistinguishable from patients in minimally conscious state.
ROLE OF THE CENTRAL THALAMUS IN SEVERE BRAIN INJURIES
Recent studies have yielded evidence for common anatomic pathologies following severe injuries associated with vegetative state10 and minimally conscious state11 as well as pathologies underlying severe to moderate cognitive disability.12 Autopsy studies of patients remaining in vegetative state at the time of death have identified widespread neuronal death throughout the thalamus as the common finding following either anoxia or diffuse axonal injury that produces widespread disruption of white matter connections.10 The severe bilateral thalamic damage after either trauma or anoxia seen in permanent vegetative state is not, however, invariably associated with diffuse neocortical neuronal cell death. This is particularly true of traumatic brain injury, in which only approximately 10% of brains at autopsy show widespread neocortical cell death.10 Specific subnuclei of the thalamus show the greatest neuronal cell loss following global and multifocal cerebral injuries produced by traumatic brain injuries.13 In particular, the central thalamic nuclei (intralaminar nuclei and related paralaminar nuclei) demonstrate progressive neuronal loss following severe traumatic brain injuries,13 and there is some evidence that a similar pattern might be identified in hypoxic-ischemic injuries.14
Progressively severe disability grades with neuronal loss along a rostrocaudal axis: the anterior intralaminar and surrounding regions initially show volume loss associated with moderate disability, while neuronal loss in the ventral and lateral nuclei of the central thalamus (posterior intralaminar group) appears with worsening disability associated with minimally conscious state and vegetative state.13 This progressive and relatively specific involvement of the nuclei of the central thalamus likely results from the unique geometry of these neurons, which have wide point-to-point connectivity across the cerebral hemisphere.15,16 The marked neuronal volume loss in these cells is likely due to their integration of the effects of neuronal cell death across large cerebral territories after diffuse trauma, hypoxia, and other nonselective severe brain injuries.
Importantly, however, focal bilateral injuries to these regions of the central thalamus are also associated with global disorders of consciousness (coma, vegetative state, and minimally conscious state).5,17 This observation indicates that these neurons also play a causal role in the production of disorders of consciousness. Abrupt injuries of the central thalamus on both sides of the brain are associated with acute coma, reflecting these cells’ key contribution to normal mechanisms of arousal regulation (reviewed by Schiff18). The central thalamus receives ascending projections from the brainstem/basal forebrain “arousal systems” that control the activity of many cortical and thalamic neurons during the sleep-wake cycle. Importantly, the central thalamus is strongly innervated by the cholinergic, serotoninergic, and noradrenergic afferents of the brainstem arousal systems (see Schiff18 for review). These same neurons also are innervated by descending projections from frontal cortical regions supporting “executive” functions that underlie goal-directed behaviors. Collectively, these ascending and descending influences on the central thalamus appear to modulate the level of arousal associated with generalized alertness and variations in cognitive effort, stress, sleep deprivation, and other variables affecting the wakeful state.15,18–22
Neuroimaging and electrophysiologic studies offer further evidence that the anatomic specializations of the central thalamus play an important role in regulating brain activation during attentive wakefulness. The central thalamus shows selective activation in normal subjects performing tasks requiring a short-term shift of attention,19,23 sustained cognitive demands of high vigilance,22 or memory holds over extended time periods.23,24 Central thalamic activation associated with varying levels of vigilance correlates with global cerebral blood flow19 and specifically covaries within the anterior cingulate cortex and pontomesencephalon. 22 Brain activity in the anterior cingulate cortex grades with increasing cognitive load and is recruited by a wide range of cognitive tasks, apparently reciprocally increasing activity along with the central thalamus in response to increasing demands of cognitive effort.20,22