Recovery of consciousness after severe brain injury: The role of arousal regulation mechanisms and some speculation on the heart-brain interface
ABSTRACT
Recovery of consciousness after severe brain injury involves reconstitution of brain arousal mechanisms and cerebral integrative function. This review discusses several aspects of neuroanatomy and neuropathology relevant to the process of recovery. Particular emphasis is placed on the role of the anterior forebrain and circuit mechanisms linking the frontal lobe, striatum, and central thalamus. The article concludes with some observations on the heart-brain interface and future research directions in the context of recovery from severe brain injury.
CIRCUIT MECHANISMS UNDERLYING RECOVERY AFTER SEVERE BRAIN INJURY
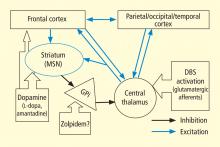
In addition to the wide point-to-point connections of the central thalamus with the cerebral cortex (predominantly connections to frontal and prefrontal cortices; see Van der Werf et al,15 Groenewegen and Berendse,28 Morel et al29), these neurons have important projections to the striatum that return via projections from the globus pallidus. 30 These projections from the central thalamus (both central lateral nucleus and parafascicularis nucleus) diffusely innervate the striatum and project onto the medium spiny neurons (MSNs), the output neuron of structure.31 Because the specific thalamostriatal projections from these central thalamic neurons use glutamate transmitters with a high probability of synaptic release,32 they likely also have a strong role in modulating background activity in the striatum.
The MSNs represent an important point of vulnerability in this anterior forebrain mesocircuit, as they have a key role in maintaining activity in the anterior forebrain through their inhibitory projections to the globus pallidus interna, which in turn inhibits the central thalamus. 33 MSNs have instrinsic cell membrane properties that keep them below their firing threshold unless a high level of spontaneous background synaptic activity arising from excitatory corticostriatal and thalamostriatal inputs is present in concert with sufficient concentrations of the neurotransmitter dopamine.33 In the setting of diffuse deafferentation or neuronal loss following severe brain injury of any type, it is expected that background excitatory synaptic activity is considerably reduced. Under these circumstances, a broad withdrawal of direct excitatory striatal projections from the central thalamus and corticostriatal inputs is likely to cause MSN output to shut down. Observations of regional changes in brain metabolism following severe brain injuries, specific responses to pharmacologic and electrophysiologic interventions in brain-injured subjects, and normal variations in brain state are all consistent with this mesocircuit model (see Schiff26 for comprehensive review). Similarly, a consistent pattern of selective metabolic downregulation within the anterior forebrain has been shown to specifically grade with severity of behavioral impairment following diffuse axonal injury,34 and application of dopaminergic agents in such patients often will produce behavioral facilitation.35,36 These medications may facilitate the output of the MSNs and directly modulate mesial frontal cortical neurons, possibly restoring anterior forebrain activity within the loop connections of the frontal cortex, striatum, pallidum, and central thalamus.
This model provides context for understanding another paradoxical observation—ie, the association of the sedative zolpidem (Ambien), a nonbenzodiazepine hypnotic that potentiates GABAA receptors, with behavioral improvement of alertness and interactive behavior in severely brain-injured patients.37–41 Zolpidem’s primary direct action in patient responders, as originally proposed by Schiff and Posner,27 may be upon on the globus pallidus interna, producing a release of tonic inhibition of the central thalamus in the setting of a broad reduction in background excitatory neurotransmission (as seen, for example, following diffuse hypoxic-ischemic injury) and leading to a shutdown of the inhibitory projection of the MSNs. The GABAA alpha-1 subunit is expressed in large quantity in the globus pallidus interna, and experimental studies support this mechanism of action.42
SINGLE-SUBJECT STUDY OF CENTRAL THALAMIC STIMULATION IN MINIMALLY CONSCIOUS STATE
A further implication of the mesocircuit model is that direct activation of the central thalamus is expected to be the causal step in reactivating a downregulated anterior forebrain system, suggesting that direct modulation of the central thalamus might facilitate behavioral responsiveness in some patients with severe brain injuries. A recent study offers evidence that direct electrical stimulation of the central thalamus can produce behavioral facilitation.
In this single-subject study of central thalamic deep brain stimulation (DBS), a 38-year-old man remained in minimally conscious state for 6 years following a severe closed head injury following blunt trauma to the right frontal lobe.43 After 3 months in a vegetative state, the patient exhibited the first evidence of clear behaviors in response to sensory stimulation consistent with minimally conscious state and advanced to eventually demonstrating a best behavioral response of inconsistent command-following and communication using eye movements. This behavioral level remained unchanged at the start of the DBS study 4 years later, as confirmed by evaluation with the Coma Recovery Scale–Revised (CRS-R), a formal behavioral assessment tool.
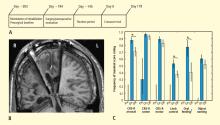
Figure 3C summarizes results of the alternating crossover study and compares the prestimulation baseline assessments of various behaviors with the “on” versus “off” testing of the DBS electrodes during the crossover phase. The results demonstrate the overall impact of DBS compared with approximately 6 months of ongoing rehabilitation efforts in the absence of DBS exposure. Overall the findings show marked improvement in behavioral responsiveness compared with prestimulation frequencies of the highest-level behavioral response across six categories. The primary outcome assessments were prospectively chosen from subscales of the CRS-R, which is a well-validated psychometric tool used in patients with disorders of consciousness. CRS-R subscale items that had shown variation during the presurgical baseline assessment were chosen prospectively as the primary outcome measures. Notably, the CRS-R oral motor subscale was not chosen because no variation in this measure had been identified during the baseline assessment period. In addition, an object-naming scale and two other tailored secondary measures were developed later, during the titration phase, as the patient’s behavior changed, and were calibrated to be tested using these secondary measurement scales. All six measures showed marked change from prestimulation baseline levels, with five of the six measures showing higher-level behaviors than those seen prior to stimulation, regardless of whether the electrodes were on or off.43
As shown in Figure 3C, the behaviors captured by secondary measures had never occurred before the titration phase of the study; ie, the patient initially lacked a capacity for object naming, oral feeding, and the complex controlled goal-directed movements captured in the secondary limb movement measure, thus setting a prestimulation baseline frequency of 0 for these measures (see supplementary material in Schiff et al43). Three outcome measures—one primary (CRS-R arousal subscale) and two secondary (oral feeding and limb control)—showed statistically significant dependence on DBS during the 6-month period, as indicated by a significantly higher frequency of maximal score rating during “on” versus “off” periods (Figure 3C). The continuation of improvements during the “off” periods of the crossover trial (relative to the prestimulation baseline assessments) showed that the DBS effects produced carryover changes that remained after the extensive exposure to DBS during the titration period (for further analysis of the dynamic of these data, see supplementary material in Schiff et al43).
Importantly, these observations are limited to a single human subject and do not provide a guide to their generalizability, 44,45 although they are consistent with the proposed mesocircuit model reviewed above. While the precise mechanism underlying this patient’s improved behavioral responsiveness with central thalamic DBS is unknown, it is likely that DBS served to partially reverse the markedly depressed cerebral global metabolism earlier measured in this patient using fluorodeoxyglucose position emission tomography (FDG-PET)46 and also seen in other patients in minimally conscious state.47 The depressed cerebral metabolism seen in minimally conscious state likely reflects volume loss of neurons, deafferentation of remaining neurons, and neuronal functional impairments. All of these mechanisms may result in low firing rates of neurons in the neocortex, thalamus, and striatum. The mesocircuit model in Figure 2 suggests that direct activation of the central thalamus in patients with such chronically downregulated background synaptic activity may produce excitatory output from central thalamic neurons that acts to partially normalize firing rates and possibly firing patterns within the corticostriatopallidal-thalamocortical system.