The American Heart Association science advisory on depression and coronary heart disease: An exploration of the issues raised
ABSTRACT
The American Heart Association issued a science advisory on depression and coronary heart disease (CHD) in 2008. This paper reviews the purpose and content of the advisory and discusses reactions and new information that have followed the advisory’s release. Both the advisory and subsequent data support routine screening for depression in patients with CHD. Such screening can be done efficiently in primary care and cardiology settings and can effectively identify many depressed patients who would otherwise go undetected. Antidepressant drugs such as selective serotonin reuptake inhibitors are safe for use in patients with CHD, can reduce depression, and can improve adherence with medical therapy. Referral to a practice with the knowledge and resources to manage depression promotes successful management of depressed patients with CHD.
In 2008, the American Heart Association (AHA) published a science advisory on depression and coronary heart disease (CHD).1 Since its publication, the advisory has evoked substantial commentary. The purpose of this article is threefold: (1) to explain the aims of the AHA science advisory, (2) to briefly discuss its content, and (3) to examine some of the comments it has provoked.
WHAT THE ADVISORY SET OUT TO DO
The purpose of an AHA science advisory is to provide rapid, clear, and consistent AHA positioning on a scientific issue. Advisories are statements on an evolving, prominent scientific issue of great interest to the public and health professionals. All AHA science advisories undergo peer review and are also reviewed and approved by the AHA Science Advisory and Coordinating Committee, AHA’s highest science body. Because this particular advisory addressed the interaction of cardiovascular and mental health, the AHA asked the American Psychiatric Association (APA) to review the document; the APA endorsed the AHA advisory.
Two points are worth emphasizing:
- An AHA science advisory is not a treatment guideline.
- Advisories usually are brief and therefore do not exhaustively discuss their topic.
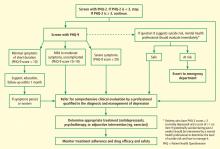
After discussing epidemiologic studies that elucidated the relationships between depression and CHD, the AHA advisory on depression and CHD focuses on screening, referral, and treatment of depression from a cardiology perspective.
The 1-year prevalence of major depressive disorder in the US general population is 7%, and the lifetime prevalence is about 16%.2 Depression in otherwise healthy persons almost doubles the risk of developing CHD.3 About 20% of patients hospitalized for acute coronary syndromes (ACS) have major depressive disorder on admission or within a few weeks thereafter, and these patients have about 2.5 times the mortality rate as patients who are not depressed, after adjusting for infarct severity and cardiovascular risk factors.4–6
ASSESSMENT OF DEPRESSION AND DEPRESSIVE SYMPTOMS
The AHA advisory discusses use of the 2-question Patient Health Questionnaire (PHQ-2) as the first step in screening for depression.7,8 The PHQ-2 inquires about the frequency of depressed mood and anhedonia by asking the following:
Over the past 2 weeks, how often have you been bothered by either of the following problems?
(1) Little interest or pleasure in doing things
(2) Feeling down, depressed, or hopeless.
For each item the response options are “not at all” (scored as 0), “several days” (scored as 1), “more than half the days” (scored as 2), and “nearly every day” (scored as 3). Thus, the total PHQ-2 score can range from 0 to 6.
Using a structured psychiatric interview as the standard, a total PHQ-2 score of 3 or greater has been shown to have a sensitivity of 83% and a specificity of 92% for major depression.7 A PHQ-2 score of 3 is the optimal cut point for screening purposes. A PHQ-2 score of 0 virtually excludes depression.
If a patient’s PHQ-2 score is 3 or greater, it is recommended that answers be obtained for a full 9-item PHQ. The PHQ-9 provides the sensitivity and specificity suitable for assigning a provisional diagnosis of major depressive disorder and a symptom severity score that can be used to identify patients for further evaluation and to make decisions about therapy.9–11
The US Preventive Services Task Force (USPSTF) currently recommends that clinicians screen adults for depression in clinical practices that have systems in place to assure accurate diagnosis, effective treatment, and follow-up.12 The USPSTF concluded that there is good evidence that screening improves the accurate identification of depressed patients in primary care settings and that treating depressed adults identified in such settings reduces clinical morbidity. Results of trials that have directly evaluated the effect of screening on clinical outcomes depend on follow-up. Limited benefits have been demonstrated in studies that simply feed screening results back to clinicians. Larger benefits have been seen in studies in which the communication of screening results is coordinated with effective follow-up and treatment. The USPSTF concluded that the benefits of screening and treating are likely to outweigh any potential harms.12
DEPRESSION TREATMENT
Drug therapy
The safety of fluoxetine, sertraline (SADHART, ENRICHD), citalopram (CREATE), and mirtazapine (MIND-IT) has been evaluated in clinical trials.4,17–21 By far the strongest evidence of safety is for selective serotonin reuptake inhibitors (SSRIs), especially sertraline.
The Sertraline Antidepressant Heart Attack Randomized Trial (SADHART; N = 369), a randomized study of depression after ACS, found no difference in cardiovascular adverse events between the sertraline and placebo groups after 16 weeks of therapy, and there were no significant differences in left ventricular ejection fraction, heart rate, blood pressure, ventricular premature complexes, or electrocardiogram changes.17 In the group assigned to sertraline, life-threatening cardiovascular events occurred less frequently (15% vs 22%, P = NS).
The Enhancing Recovery in Coronary Heart Disease study (ENRICHD) was a large trial (N = 2,481) to evaluate the effect of cognitive behavioral therapy (CBT) on depression or low perceived social support in patients enrolled within 28 days of myocardial infarction (MI).4 The ENRICHD protocol required patients randomized to CBT with Hamilton Depression Rating Scale (HAM-D) scores greater than 24, or who showed less than 50% reduction in Beck Depression Inventory (BDI) scores after 5 weeks of CBT, to be referred to a study psychiatrist for consideration of pharmacotherapy, usually sertraline. Of the overall ENRICHD population, 1,834 participants (74%) had a diagnosis of depression, and 446 of these participants (24%) were treated with antidepressant drugs, 301 with an SSRI and 145 with other antidepressants.18 During mean follow-up of 29 months, the SSRI-treated group had a statistically significant 43% reduction in death or MI.4
The Canadian Cardiac Randomized Evaluation of Antidepressant and Psychotherapy Efficacy (CREATE) recruited 284 patients with chronic CHD, major depressive disorder, and a 24-item HAM-D score of 20 or greater and randomly assigned half to citalopram (another SSRI) and half to placebo.19 Citalopram showed antidepressant efficacy and no evidence of harm.
The Myocardial Infarction and Depression Intervention Trial (MIND-IT) recruited 91 patients within 30 days of hospital admission for MI with depression and randomized them in a 1:1 ratio to mirtazapine or placebo. Mirtazapine showed some evidence of antidepressant efficacy and no evidence of harm.20
Strik et al in the Netherlands recruited 54 patients who had major depression after a first MI and randomized them 1:1 to the SSRI fluoxetine or placebo 3 months after the MI.21 The fluoxetine group showed a trend toward antidepressant efficacy, and no cardiovascular safety problems were detected clinically or by either electrocardiogram or echocardiogram.