Neuromodulation of cardiac pain and cerebral vasculature: Neural mechanisms
ABSTRACT
Research using animal models has helped elucidate the neural mechanisms of angina pectoris, sensitization of cardiac nociceptive stimuli, and neuromodulation of cardiac pain and cardiovascular function. Findings over the last 2 decades include evidence of convergence of visceral-somatic input to spinothalamic cells and a major role for the vagus nerve in spinal cord processing. Stress-related glucocorticoids may manipulate amygdala function, inducing hypersensitivity to nociceptive input from the heart via central sensitization of upper thoracic spinal neuronal activity. Spinal cord stimulation may have therapeutic effects, although the underlying mechanism is unclear.
NEUROMODULATION OF CEREBROVASCULATURE AND CARDIAC PAIN
Neuromodulation of cerebral blood flow
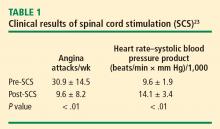
Spinal cord stimulation is used to treat several cerebrovascular disorders, including cerebral ischemia, focal cerebral ischemia, stroke, postapoplectic spastic hemiplegia, and prolonged coma (see Yang et al13 for citations that address these pathologies). There is no clear explanation for its therapeutic effect; mechanisms being investigated include changes in cerebral blood flow and processing of nociceptive information.
To assess the effect of spinal cord stimulation on cerebral blood flow, we exposed the C1–C2 area of an anesthetized rat, stimulated the area with a ball electrode, and used laser Doppler flow probes to measure the blood flow on the surface of the cortex bilaterally. 13 The stimulus parameters were 30%, 60%, and 90% of motor threshold; the threshold was determined by gradually increasing the intensity of spinal cord stimulation until the neck muscles contracted. Blood flow increased on both sides with increasing stimulation intensities.13
Other studies have evaluated cerebral blood flow but did not measure change in cerebrovascular resistance. We observed that spinal cord stimulation—particularly at 60% and 90% of motor threshold—increased blood flow and reduced resistance to spinal cord stimulation on the dorsal columns at C1, both ipsilaterally and contralaterally.
In other tests, cerebral blood flow and vascular resistance to spinal cord stimulation were not changed after transection of the spinal cord at the C6–C7 segments. These results suggested that information was not being transmitted to the sympathetic nervous system via the thoracic spinal cord. We applied ibotenic acid to C1–C2 to assess whether the underlying stimulated neurons affected cerebral blood flow; there was no significant change. On the other hand, a small cut in the dorsal column rostral to the stimulation site caused significantly reduced cerebral blood flow and vascular resistance, indicating that the dorsal columns function in an ascending manner to produce the vasodilation in the cerebral cortex.13
Capsaicin-sensitive sensory nerves, which contain transient receptor potential vanilloid-1 (TRPV1) receptors, may have a role in spinal cord stimulation–induced vasodilation. TRPV1 receptors are nonselective cation channels activated by capsaicin, heat, and hydrogen ions.14 Activation, which causes an influx of cations and release of calcitonin gene-related peptide (CGRP) and substance P, is related to the pathogenesis of inflammation and hypertension. To examine the potential role played by capsaicin-sensitive sensory nerves, we administered resiniferatoxin (RTX), an ultrapotent capsaicin agonist; RTX specifically targets and desensitizes TRPV1-containing sensory fibers.13,15 Administration either intravenously or by direct application to the spinal cord results in a 15- to 20-minute period of sensitization followed by several hours of desensitization; if exposure lasts for several days, the nerves are destroyed.
Intrathecal administration of RTX to the spinal cord resulted in no significant change in cerebral blood flow. However, intravenous administration resulted in significantly decreased cerebral blood flow and decreased resistance, suggesting a role for TRPV1 receptors in cerebral blood flow.13
There may be a connection between spinal cord stimulation at C1 and vasodilation of the cortex. The literature suggests that spinal cord stimulation activates the dorsal column nuclei16; we found evidence of this in our laboratory when we recorded activity from cells in the cuneate and gracilus nuclei after spinal cord stimulation. There is also a possible pathway between the dorsal column, the rostral ventrolateral medulla, and the sphenopalatine ganglion that influences vasodilation.17–20 Although not yet clearly defined, evidence suggests a connection between spinal cord stimulation and transmission of this information through the dorsal columns to influence vasodilation.17–20
Neuromodulation of thoracic spinal processing of cardiac nociceptive information
Using a rat model to assess the effects of spinal stimulation, we recorded T3 activity during dorsal column stimulation of either C8-T1 or C1–C2 segments. Activity was almost completely suppressed with C1–C2 stimulation during bradykinin injection into the pericardial sac. The results suggest that spinal cord stimulation suppresses the processing of nociceptive information.24
Stimulating the spinal cord at C8-T1 also suppresses the effect of bradykinin. One possible mechanism for this effect is that spinal cord stimulation activates large afferent fibers; GABAergic connections in the superficial dorsal horn may suppress the processing of information in the spinothalamic tract neurons.22,25
SUMMARY
Our investigations have generated information about afferent input to the spinothalamic tract cells, the effects of glucocorticoids on amygdala function, and possible therapeutic mechanisms of spinal cord stimulation.
We have demonstrated convergence of viscerosomatic input in spinothalamic cells. There is virtually no viscerocardiac input at the C7–C8 region, but there is input at C5–C6. Vagal afferent activity is the major source of input at the C1–C2 region; in this region. Vagal nerve stimulation may have a major role in processing in the upper cervical spinal cord and may change the balance of processing in the supraspinal nuclei.
Glucocorticoids manipulate amygdala function by inducing hypersensitivity to nociceptive input from the heart through central sensitization of upper thoracic spinal neuronal activity. Descending information from the amygdala depends, in part, on the C1–C2 propriospinal pathway.
Spinal cord stimulation at C1–C2 or C8-T1 can activate inner neuronal mechanisms that may involve GABA, modulating the wide dynamic range of neurons that are part of the spinothalamic tract.