Bakken Lecture: The brain, the heart, and therapeutic hypothermia
ABSTRACT
Therapeutic hypothermia in acute resuscitation medicine has a long history, but its currently recommended use dates back to work in the mid-1960s by the late Dr. Peter Safar and colleagues. Compared with normothermia, mild therapeutic hypothermia, induced right after restoration of spontaneous circulation in comatose survivors of cardiac arrest, leads to 1 additional patient with intact neurological outcome for every 6 patients treated. Demonstrating benefit from therapeutic hypothermia in other acute neurological insults, such as traumatic brain injury, has been more difficult. Current research to optimize the benefits of mild therapeutic hypothermia in cardiac arrest is focused on hypothermia’s profound effects on drug metabolism, determining the best anesthetics and sedatives to use with cooling, and identifying compounds that may promote induction of hypothermia or create a poikilothermic state. Future applications of therapeutic hypothermia may include induction of emergency preservation and resuscitation to buy time for damage-control surgery in patients with exsanguination cardiac arrest.
FUTURISTIC APPLICATIONS OF THERAPEUTIC HYPOTHERMIA
Emergency preservation and resuscitation
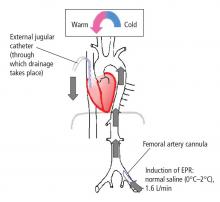
Exsanguination cardiac arrest is one of the most refractory types of cardiac arrest, with mortality rates generally greater than 95%.31 Obviously, therapies such as CPR are ineffective in the absence of an adequate circulating blood volume.
In initial reports,32–34 we targeted relatively brief insults ranging from 15 to 60 minutes. We determined that for insults at or beyond 60 minutes, profound levels of hypothermia (tympanic temperature of ~10°C) were most effective.34 In subsequent studies, we demonstrated that pharmacologic adjuncts to hypothermia were relatively ineffective. Indeed, only one agent, the brain-penetrating antioxidant Tempol, enhanced the efficacy of profound hypothermia.35 We also demonstrated that the prolonged use (36 to 48 hours) of mild hypothermia after the acute application of EPR further enhanced neurological outcomes as compared with more rapid rewarming.36 Similarly, unlike drugs, the addition of energy substrates (namely, dissolved oxygen and 2.5% dextrose) to the flush facilitated the ability to achieve remarkably long EPR durations in experimental exsanguination cardiac arrest—as long as 3 hours of preservation at approximately 10°C.37 These findings could also have important implications for optimizing conventional use of deep hypothermia circulatory arrest in cardiac or neurological surgery. We also have recently developed a rat model of EPR using a miniaturized cardiopulmonary bypass system. It is used to screen novel therapeutic adjuncts to EPR and to study mechanisms of neuroprotection in this special paradigm.38,39
Two other investigative teams, one at Harvard University and another at the Vienna General Hospital, have also been exploring the use of EPR-related technologies—and observing similar success. Alam et al40 have used a low-flow EPR approach in pigs to facilitate damage-control surgery after otherwise lethal traumatic insults. Janata et al41 have successfully used EPR in the setting of refractory normovolemic cardiac arrest, simulating the typical cardiac arrest victim who cannot be resuscitated in either the field or the emergency department.
Finally, the EPR concept recently received funding to proceed to a clinical trial in civilian trauma. The study, to be led by Dr. Samuel Tisherman, one of the pioneers of this approach at the Safar Center, will include several trauma centers in the United States and target otherwise lethally injured trauma victims with exsanguination cardiac arrest.