A 19-year-old man with progressive lung infiltrates
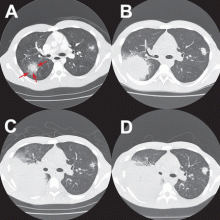
A 19-year-old man received induction chemotherapy with idarubicin and cytarabine for secondary acute myeloid leukemia. Subsequently, he developed fever, progressive lung infiltrates, and severe neutropenia. His white blood cell count was 1.1 × 109/L (reference range 4.0–11.0) with 100% lymphocytes; his blood glucose level remained normal.
The patient was treated with voriconazole (Vfend) and broad-spectrum antibiotics.
Q: Which is the most likely cause?
,- A bacterium
- A virus
- Cryptococcus
- Aspergillus
- Zygomycetes
A: The correct answer is Zygomycetes, the second most common cause of fungal respiratory disease in patients with hematologic malignancies.
UNCOMMON BUT OFTEN FATAL
Zygomycetes is a class of fungi that contains two orders, Mucorales and Entomophthorales. Human disease, which is uncommon but frequently fatal, is predominantly associated with Mucorales and is commonly called mucormycosis.1,2
The major mode of transmission is through inhalation of spores from diverse decaying environmental sources. As Zygomycetes are aerogenous pathogens, they predominantly affect the paranasal sinuses and the lungs. The main risk factors for Zygomycetes infection include diabetes mellitus, hematologic malignancies (predominantly acute leukemias treated with aggressive chemotherapeutic regimens), pharmacologic immunosuppression, solid organ or bone marrow transplantation, and therapy with deferoxamine (Desferal), an iron-chelating agent.1,3
Overall, rhinocerebral disease is the most common manifestation, especially in the setting of diabetic ketoacidosis.1 In hematologic malignancies, the most common presentation is pulmonary zygomycosis with associated profound neutropenia, as neutrophils are the central defense against filamentous fungal hyphae.1,4
The incidence of zygomycosis is increasing, likely owing to the greater number of patients receiving stem cell or solid organ transplants, the use of more aggressive immunosuppressive regimens, prolonged survival, and the frequent prophylactic use of antifungal agents without activity against Zygomycetes.2,5
INFECTION PROGRESSES RAPIDLY
Most patients present with fever, cough, thoracic pain, and dyspnea in association with hypoxemia and pulmonary infiltrates refractory to broad-spectrum antibiotics. Zygomycosis can also present radiographically as pulmonary nodules or consolidations with or without the halo sign or cavitations.6,7
The disease usually progresses rapidly, invading vessels and causing infarction, bleeding, and dissemination to extrapulmonary sites.1,5
In reported cases in patients with acute leukemia who received aggressive chemotherapy, the fungal infection occurred several days after profound neutropenia developed.3,4
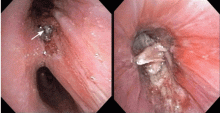
SPUTUM, LAVAGE, BIOPSY
The diagnosis is based on directly identifying the fungus morphologically and on culturing it. However, cultures of sputum, BAL fluid, and blood are usually negative.
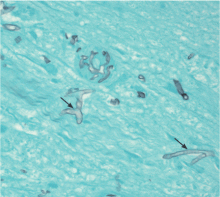
Morphologically, the fungus is broad with irregular walls; it is also nearly aseptate and frequently has right-angle branching. In contrast, Aspergillus is narrow with parallel walls, distinctive septae, and acute branching.2
Of note: physicians need to alert the microbiology laboratory about their clinical suspicion of Zygomycetes infection, because the recovery rate of Zygomycetes in culture is increased by slicing the biopsy specimen in small pieces but not dicing it (to avoid breaking the septae).1
The diagnostic tests include microscopic examination and culture of sputum, BAL fluid, and transbronchial biopsy specimens. If the initial tests are negative but the suspicion of zygomycosis is strong on clinical grounds, then fine-needle aspiration or open lung biopsy should be considered.1
Useful predictors that favor the diagnosis of pulmonary zygomycosis instead of the main alternative, invasive pulmonary aspergillosis, include concomitant sinusitis, voriconazole prophylaxis (due to antifungal pressure), a negative galactomannan test in serum, multiple pulmonary nodules, and pleural effusion.2,8