Diffuse alveolar hemorrhage: Diagnosing it and finding the cause
ABSTRACTDiffuse alveolar hemorrhage is an acute, life-threatening event, and repeated episodes can lead to organizing pneumonia, collagen deposition in small airways, and, ultimately, fibrosis. Among the many conditions it can accompany are Wegener granulomatosis, microscopic polyangiitis, Goodpasture syndrome, connective tissue disorders, antiphospholipid antibody syndrome, infectious or toxic exposures, and neoplastic conditions. Its many causes and presentations pose an important challenge to the clinician.
KEY POINTS
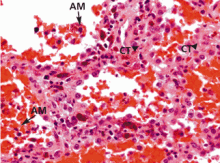
- Most patients present with dyspnea, cough, hemoptysis, and new alveolar infiltrates. Early bronchoscopy with bronchoalveolar lavage is generally required to confirm the diagnosis; blood in the lavage specimens (with numerous erythrocytes and siderophages) establishes the diagnosis.
- Therapy targets both the autoimmune destruction of the alveolar capillary membrane and the underlying condition. Corticosteroids and immunosuppressive agents remain the gold standard.
- In patients with diffuse alveolar hemorrhage and renal impairment (pulmonary-renal syndrome), kidney biopsy can be considered to identify the cause and to direct therapy.
Bronchoscopy
The diagnostic evaluation in diffuse alveolar hemorrhage usually includes bronchoscopic examination,10 which serves two purposes:
- To document alveolar hemorrhage by bronchoalveolar lavage and to exclude airway sources of bleeding by visual inspection
- To exclude an associated infection.
Based on experience with nonmassive hemoptysis of all causes (but not exclusively diffuse alveolar hemorrhage), the diagnostic yield of bronchoscopy is higher if the procedure is performed within the first 48 hours of symptoms rather than later. Evidence supporting diffuse alveolar hemorrhage is persistent (or even increasing) blood on three sequential lavage aliquots from a single affected area of the lung.
FINDING THE UNDERLYING CAUSE
Once the diagnosis of diffuse alveolar hemorrhage is established, the clinician must ascertain whether an underlying cause is present. Serologic studies may prove important, although the results are generally not available in a manner timely enough to guide immediate management.
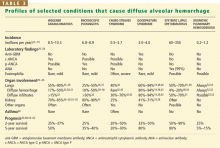
When a pulmonary-renal syndrome is suggested by accompanying hematuria or renal dysfunction, antiglomerular basement membrane antibody and antineutrophil cytoplasmic antibody (ANCA) levels should be checked. Tests for complement fractions C3 and C4, anti-double-stranded DNA, and antiphospholipid antibodies should be ordered if an underlying condition such as lupus or antiphospholipid antibody syndrome is suspected (Table 2).11
If the underlying cause remains elusive after a thorough clinical evaluation that includes imaging studies, serologic studies, and bronchoscopy, then surgical biopsy should be considered.1 Which organ to biopsy (eg, lung, sinus, kidney) depends on the level of suspicion for a specific cause. For example, suspicion of Wegener granulomato-sis with hematuria or renal dysfunction might prompt renal biopsy. However, lung biopsy often needs to be performed with video-assisted thoracoscopy, especially when disease is confined to the lung (as in idiopathic pulmonary hemosiderosis or pauci-immune pulmonary capillaritis). Renal biopsy specimens should also undergo immunofluores-cence staining, which may reveal linear deposition of immunoglobulins and immune complexes along the basement membrane in patients with Goodpasture syndrome, or of granular deposits in patients with systemic lupus erythematosus.
TWO GENERAL CLINICAL SCENARIOS
In general, the clinician will be confronted by one of two scenarios: a patient with diffuse alveolar hemorrhage and associated systemic findings, or a patient with hemorrhage and no associated systemic findings.
Hemorrhage with associated systemic findings
Certain clues from the history raise suspicion of diffuse alveolar hemorrhage:
- Recent infection suggests Henoch-Schönlein purpura or cryoglobulinemic vasculitis
- Use of a possibly offending drug such as an anticoagulant, D-penicillamine (Cuprimine, Depen), nitrofurantoin (Furadantin, Macrobid, Macrodantin), amiodarone (Cordarone), propylthiouracil, cocaine, or sirolimus (Rapamune, Rapamycin)
- Exposure to toxic agents such as trimellitic anhydride, insecticides, and pesticides
- A known comorbid condition such as vasculitis, connective tissue disease, mitral valve disease, or solid organ or stem cell transplantation.
If asthma, eosinophilia, pulmonary infiltrates, and diffuse alveolar hemorrhage coexist, consideration should be given to Churg-Strauss syndrome. If sinus disease, skin manifestations, pulmonary parenchymal nodules, and cavitary lesions coexist with positivity for antiproteinase 3 c-ANCA and biopsy-proven granulomata, then Wegener granulomatosis should be considered. Similarly, diffuse alveolar hemorrhage with glomerulonephritis and skin manifestations, positivity for p-ANCA, and necrotizing nongranulomatous lesions on end-organ biopsy may lead to a diagnosis of microscopic polyangiitis. In a young smoker with glomeru-lonephritis and diffuse alveolar hemorrhage presenting as either bland alveolar hemorrhage or pulmonary capillaritis, Goodpasture syndrome or antiglomerular basement membrane antibody disease should be considered.
Hemorrhage with no associated systemic findings
When the above conditions have been considered but no suggestive findings are found, the following four conditions should be considered:
- Antiglomerular basement membrane antibody disease in limited pulmonary form or onset: positivity to the antibody with linear deposits in the lungs would be diagnostic in such a case
- Pulmonary-limited microscopic polyangiitis positive for p-ANCA (a positive anti-myeloperoxidase p-ANCA test makes the diagnosis)
- Pauci-immune isolated pulmonary capillaritis, when the biopsy shows evidence of neutrophilic pulmonary capillaritis
- Idiopathic pulmonary hemosiderosis, a diagnosis of exclusion, when the biopsy shows evidence of acute, subacute, and chronic bland diffuse alveolar hemorrhage and no evidence of vasculitis.