Genitourinary syndrome of menopause: Common problem, effective treatments
ABSTRACT
After menopause, about half of all women experience genital, sexual, and urinary symptoms associated with decreases in estrogen, termed genitourinary syndrome of menopause. First-line therapies are nonhormonal vaginal lubricants and moisturizers. For persistent symptoms, prescription estrogen in cream and ring formulations is effective.
KEY POINTS
- Practitioners can use the patient’s most bothersome symptom and her vaginal pH level to assess clinical responses to therapy.
- The diagnosis is based on clinical signs and symptoms from the medical history and physical examination.
- If the symptoms are not bothersome to the patient, the syndrome does not require treatment.
SELECTING A TREATMENT
Symptomatic women with GSM who desire intervention should be offered over-the-counter nonhormonal products as the first line of therapy.
If nonhormonal products are ineffective and there are no contraindications, locally applied estrogen in cream, tablet, or a ring delivery system may be offered. Local dehydroepiandrosterone (DHEA) inserts or ospemifene, an oral selective estrogen-receptor modulator, are FDA-approved for moderate to severe dyspareunia secondary to GSM.
Oral estrogen therapy is not indicated for vulvovaginal symptoms, but some women taking systemic estrogen for vasomotor symptoms may need additional local estrogen application to relieve vaginal symptoms.
Nonhormonal treatments
Nonhormonal over-the-counter therapies provide sufficient relief for most women with mild symptoms. There is a plethora of products, so practitioners need to offer guidance to help women with their individual choices.
Vaginal lubricants are intended for use with sexual or penetrative activity (including pelvic examination). They provide short-term relief of symptoms, but there is no evidence of any impact on histologic changes of atrophy. They are meant to relieve friction. Lubricants may be water-based, oil-based, silicone-based, or a combination. Individual products have different effects on condom integrity. Perfumed, warming, or stimulating products may be irritating to some women and should be tried initially in small amounts.
Vaginal moisturizers are intended to treat GSM. They are applied regularly, not just with vaginal activity, usually once or twice a week. Some vaginal lubricants can maintain an acidic pH in the vagina and may reverse the histologic changes of atrophy. Symptomatic improvement over placebo or estrogen has been shown in clinical trials.22–24
Women should be advised that trial and error in choosing products may be necessary to establish a successful regimen. Products should be tried in succession, not simultaneously, with a “wash-out” period between, to be able to evaluate response.
Vaginal dilators and pelvic floor physical therapy
Sexual activity, either by self-stimulation or with a partner, helps maintain vaginal health by contributing to increased vascularity and elasticity of tissue. Women who resume sexual activity after a long period of inactivity may benefit from the use of vaginal dilators, which aid both in mechanical distention and progressive relaxation of the vaginal musculature.
In some women, long-term dyspareunia may result in vaginismus, an involuntary contraction of the vaginal musculature. For these women, dilators may be effective. Additional options focus on pelvic floor physical therapy, which can isolate trigger points, using biofeedback to teach relaxation and home exercises such as vaginal massage.
HORMONAL THERAPIES
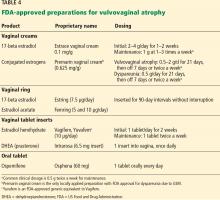
If nonhormonal lubricants and moisturizers do not achieve satisfactory symptomatic relief, FDA-approved hormonal therapies (Table 4) include estrogen-containing vaginal creams, rings, and a tablet; a vaginal tablet containing DHEA; and an oral tablet containing ospemifene.
Estrogen products
For patients whose symptoms do not respond to nonhormonal therapies, low-dose, locally applied estrogen therapy is the first treatment recommended.2 Locally applied estrogens can reverse the atrophic changes of estrogen deprivation, resulting in an increase in blood flow, elasticity, and vaginal wall thickness. This therapy also can normalize pH levels with subsequent restoration of a healthy lactobacilli-based flora. Locally applied estrogens also have been shown to decrease the frequency of recurrent urinary tract infection.25
Estrogen-containing vaginal creams, rings, and a tablet are available, and each has been shown to be effective for GSM. Locally applied estrogens at recommended dosages tend to have fewer adverse events and risks than systemic estrogens.26 Estradiol levels generally do not exceed levels found in the untreated menopausal population, although a dose- and duration-dependent increase in systemic levels may occur.27
Dosing considerations
The vaginal ring and the vaginal tablet provide the lowest prefixed daily dose of estradiol (7.5 and 10 µg daily, respectively). Estrogen creams (estradiol, conjugated equine estrogens) are more readily absorbed, and dosing should be tapered to the lowest, most effective dose for symptom relief.
The FDA-approved doses for vaginal creams containing 17-beta estradiol are higher than the dose found to be effective in clinical practice (0.5 g twice a week). Most practitioners start with the lower dose, reserving the FDA-approved higher doses for patients who do not obtain adequate relief over 6 to 8 weeks of treatment. The conjugated-estrogen vaginal cream Premarin is the only locally applied estrogen approved by the FDA to treat dyspareunia. It is dosed at 0.5 g intravaginally for 21 days and is then either withdrawn for 7 days or, more commonly, administered at 0.5 g twice a week.
Initial treatment with vaginal cream may require more frequent vulvovaginal application, such as daily for 1 to 2 weeks. Women with vaginal fissures or tearing will benefit from externally applied creams in addition to internal applications. Response to therapy is usually seen within 4 to 6 weeks from onset of treatment. Once symptom relief is obtained, treatment should continue indefinitely. Although long-term safety studies are lacking, risks are believed to be minimal.
Endometrial impact. Women with contraindications to systemic estrogen should be counseled about possible small increases in serum levels of estradiol associated with locally applied estrogens and the potential risks and benefits those increases incur. Endometrial surveillance with either transvaginal ultrasonography or endometrial sampling is not required, even with long-term use, but it should be considered with higher doses or more frequent applications.
Similarly, progesterone replacement for endometrial protection is not recommended but can be considered in women with an intact uterus at high risk of endometrial cancer, such as obese patients. If a systemic progestational agent is considered, the risks and benefits should be weighed carefully. Even in women at high risk, endometrial surveillance may be the most appropriate option.28 Uterine bleeding that occurs should be considered abnormal and should be investigated.
DHEA (prasterone)
In 2016, the FDA approved intravaginal prasterone, a DHEA-containing product for the treatment of dyspareunia secondary to moderate to severe vulvovaginal atrophy caused by menopause. DHEA is an endogenous steroid that is converted by aromatase activity into testosterone and estradiol.
Clinical trials have found that 12 weeks of vaginal DHEA supplementation (0.25%, 0.5%, and 1% DHEA ovules) was more effective than placebo in improving vaginal dryness and dyspareunia in women with GSM.29–31 In these studies, locally applied DHEA decreased parabasal cells, decreased vaginal pH, increased vaginal secretions, and improved epithelial surface thickness and integrity without any significant impact on serum levels of DHEA, DHEA-sulfate, estradiol, testosterone, or their metabolites. Importantly, transvaginal DHEA had negligible endometrial effect.
The breast cancer risk associated with vaginal DHEA has not been fully evaluated. However, labeling lists breast cancer as a warning, not a contraindication.