Patient and treatment perspectives: Revisiting the link between type 2 diabetes, weight gain, and cardiovascular risk
ABSTRACT
Lifestyle modifications in conjunction with antidiabetes medications can produce near-normal blood glucose concentrations in patients with type 2 diabetes mellitus (T2DM). Because these patients have increased cardiovascular morbidity and mortality, treatment strategies should also address the cardiovascular aspects of the disease, including blood pressure, lipids, and body weight. Since the prevalence of these abnormalities is increasingly secondary to poor diet and sedentary lifestyles and because most patients with T2DM are overweight/obese, clinicians are encouraged to help patients reduce body weight while correcting hyperglycemia by selecting treatment options that improve both parameters. The glucose-lowering properties of insulin and sulfonylureas are well known but they are also associated with weight gain. Thiazolidinediones are associated with weight gain as well as edema. However, this weight gain may be more peripheral than central, which may mitigate the risks associated with increased body fat. Metformin, the consensus first-line drug for the treatment of patients with T2DM, is weight neutral. Newer antidiabetes agents include incretin-based medications, such as the glucagon-like peptide–1 receptor agonists, which tend to decrease weight, and the dipeptidyl peptidase–4 inhibitors, which are weight neutral.
KEY POINTS
- Control of cardiovascular risk factors is as important as glycemic control in patients with T2DM.
- Intensive glucose control has shown mixed results in terms of correlation with improved cardiovascular risk factors.
- Newer agents target the fundamental pathophysiologic defects of T2DM, with beneficial effects on weight and other cardiovascular risk factors.
PATIENT ADHERENCE AND SATISFACTION
It is often challenging for patients with T2DM to adhere to their treatment regimens. The Diabetes Attitude, Wishes, and Needs (DAWN) study examined psychosocial barriers to self-care in patients with diabetes and found that while 78% of patients with T2DM adhered to their medications, only 39% achieved complete success in at least two-thirds of their self-care domains.19 A multicenter, randomized, clinical trial examined the correlates of treatment satisfaction, including body weight, on patients’ appraisal of treatment satisfaction with injectable insulin. The 14.5% of patients who experienced a reduction in BMI reported systematic improvement in treatment satisfaction.20 Similarly, a cross-sectionally designed study (n = 99) that analyzed the interrelation of adherence, BMI, and depression in adults with T2DM found that patients with higher BMI and poor adherence also had depression, which was mediated by lower self-efficacy perceptions and increased diabetes symptoms.21 The results from these studies show a clear relationship between adherence with treatment regimens and achievement of HbA1c goals.22
RECENT DEVELOPMENTS IN T2DM MANAGEMENT: STRATEGIES TO REDUCE CV RISK
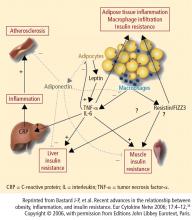
Because excess weight and obesity are prominent features of T2DM, it is important to use an antidiabetes agent that does not induce unnecessary weight gain (particularly central weight gain, which is thought to be most atherogenic).23 Metformin, considered the first-line agent for treatment of T2DM, is generally weight neutral with a low level of hypoglycemia.24,25 Sulfonylureas, insulin, and thiazolidinediones (TZDs) are all associated with weight gain, although newer-analogue insulins may cause less weight gain than older agents. TZDs, especially pioglitazone, are associated with improvements in long-term beta-cell function and CV risk factors despite weight gain.26,27
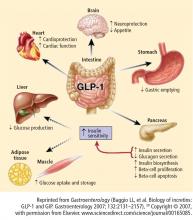
The newer antidiabetes agents belong to the dipeptidyl peptidase–4 (DPP-4) inhibitor and the glucagon-like peptide–1 (GLP-1) receptor agonist therapeutic classes and have been shown to be either weight neutral (DPP-4 inhibitors) or to cause weight loss (GLP-1 receptor agonists).28
Obesity and the incretin effect
Two recent studies showed that surgically induced weight loss enhances the physiologic “incretin effect.” In one study, obese individuals with T2DM whose weight loss was secondary to bariatric surgery combined with caloric restriction showed improved insulin sensitivity, improved carbohydrate metabolism, and elevated levels of adiponectin and GLP-1, all of which may reduce the incidence of T2DM.36 In the other study, bariatric surgery in morbidly obese individuals with T2DM improved insulin secretion and ameliorated insulin resistance.37
DPP-4 inhibitors
DPP-4 inhibitors such as sitagliptin and saxagliptin inhibit the enzymatic activity of DPP-4 and increase endogenous concentrations of GLP-1.28 Sitagliptin has been compared with placebo as monotherapy and has been studied in combination with other therapies.
In an 18-week study, sitagliptin monotherapy, 100 and 200 mg QD, significantly reduced HbA1c compared with placebo (placebo-subtracted HbA1c reduction, –0.60% and –0.48%, respectively) in patients with T2DM. Sitagliptin also significantly decreased fasting plasma glucose (FPG) concentration relative to placebo.38 Twelve weeks of sitagliptin monotherapy at dosages of 5, 12.5, 25, and 50 mg BID led to significant (P < .001) reductions in HbA1c compared with placebo. Sitagliptin also produced significant reductions in FPG and mean daily glucose concentrations across the doses studied.39 Similar results were reported in other 12-week studies: 50 mg BID and 100 mg QD sitagliptin monotherapy significantly (P < .05) reduced HbA1c –0.39% to –0.56% and FPG concentration –11.0 to –17.2 mg/dL compared with placebo40; sitagliptin 100 mg QD compared with placebo produced a least-squares mean change from baseline HbA1c of –0.65% versus 0.41% (P < .001) and FPG of –22.5 versus 9.4 mg/dL (P < .001).41
Sitagliptin also has been studied in combination with other therapies. After 24 weeks, sitagliptin combined with pioglitazone significantly reduced HbA1c by –0.70% and FPG by –17.7 mg/dL (P < .001 for both) compared with placebo.42 In another 24-week study, 100 mg sitagliptin QD significantly improved glycemic control and beta-cell function (P < .05 for both) in patients with T2DM who had inadequate glycemic control with glimepiride or glimepiride plus metformin.43
In addition to significantly reducing HbA1c, sitagliptin 100 and 200 mg QD produced only small differences in body weight relative to placebo: least-squares mean change from baseline for sitagliptin 100 mg was –0.7 kg (95% CI, –1.3 to –0.1) and for 200 mg was –0.6 kg (95% CI, –1.0 to –0.2); for placebo it was –0.2 kg (95% CI, –0.7 to 0.2).38 These findings were consistent with those from another 24-week monotherapy study where sitagliptin produced weight loss of up to –0.2 kg44 and a 30-week study of sitagliptin added to ongoing metformin therapy. In the latter study, both sitagliptin and placebo resulted in weight reductions of –0.5 kg.45
The effects of sitagliptin on lipids and BP have been reported in clinical studies in patients with and without T2DM. In one study of patients with T2DM, the addition of sitagliptin to metformin increased total cholesterol (+8.1 mg/dL), LDL-C (+9.2 mg/dL), and HDL-C (+1.8 mg/dL) but lowered triglyceride (–14.5 mg/dL) after 18 weeks of treatment (24-week data).46 Data from a small (n = 19) study in nondiabetic patients with mild to moderate hypertension showed that sitagliptin produced small reductions (–2 to –3 mm Hg) in 24-hour ambulatory BP measurements.47
Another DPP-4 inhibitor, saxagliptin, with efficacy similar to that described for sitagliptin, was recently approved by the US Food and Drug Administration (FDA) for treatment of T2DM.48