Considerations for Optimal Inhaler Device Selection in Chronic Obstructive Pulmonary Disease
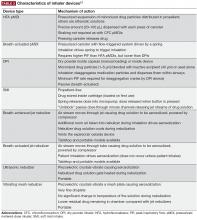
Device considerations
The SMI delivers the aerosol as a fine mist with slow velocity lasting >1 second, which is considerably slower than spray delivery with pMDIs.14 The aim of this design is to make it easier for patients to coordinate actuation with inhalation, but it is important to note that some coordination is still required for SMI devices to function correctly.14 In addition, the SMI is not dependent on a patient’s ability to generate sufficient PIF for effective drug delivery. A limitation of the SMI is the need to assemble the device, as patients with poor manual dexterity may encounter difficulty when attempting to load the drug cartridge.15
Nebulizers deliver aerosolized drug in a fine mist. Newer-generation portable vibrating mesh nebulizers can deliver a dose over a period of ~2 minutes, compared with 10 minutes for conventional pneumatic devices.16 Patients find them effective and easy to use, and the newer generation devices overcome problems with portability and length of treatment, which may be an issue during the daytime for ambulatory patients, along with the requirement for cleaning after each dose.4,8 However, drug delivery may be somewhat compromised with nebulizers compared with other inhalation devices, as medication can be dispersed into the atmosphere and lost, rather than inhaled.7 An additional point to consider is medication availability; some medications, particularly fixed-dose combination maintenance therapies, are currently unavailable in a nebulized format.16
The most important device-related factors influencing the site of deposition within the lungs are aerosol velocity and particle size of the inhaled drug.3,7,17 To maximize clinical effectiveness, adequate distribution throughout the lung is required to reach target sites of action for β2-agonists, anticholinergics, and corticosteroids.17 Particle size differs between inhaler device types, but all available devices generate drug particles sufficient for deposition throughout the lower airways and lung periphery, ie, within the range of 1–5 microns.3,18-21 Extra fine particles of <1 micron (or “submicron particles”) can be deposited deeper in the pulmonary acinus, but a higher fraction of such particles may be exhaled compared with particles 1–5 microns in size.3,20,22 In contrast, particles >5 microns deposit in the oropharynx and may be swallowed, potentially leading to systemic adverse effects.3,20,22
When more than one drug is required, it may be preferable to deliver them via a single device where possible to facilitate patient compliance with correct technique, and decrease confusion about how to use different inhalers.23 The inhaler device ideally serves as a platform on which many treatments are available; the greater the number of devices employed by the patient, the greater the likelihood of making an error with the usage of each device.24