Cardiac implantable electronic device infection
ABSTRACT
Increasing numbers of patients with cardiac disease have improved quality of life and longevity as a result of cardiac implantable electronic devices (CIEDs). CIED infections can involve the generator pocket, bloodstream, or cardiac structures and occur in about 0.5% of de novo CIED implants and approximately 2% of CIED replacements. Prompt diagnosis of CIED infection is beneficial to the success of antibiotic therapy and subsequent device removal to resolve the infection. Measures to prevent CIED infections include assessment of the indication and patient status, strict sterile surgical techniques, preoperative antibiotics, and adequate homeostasis. New surgical methods and CIED devices may also lead to reduction in CIED infections. Further research is needed to better quantify the incidence of CIED, risk factors, and efficacy of surgical techniques to prevent infections.
KEY POINTS
- CIED use is increasing, as are the number of CIED infections, which are associated with significant morbidity and mortality.
- Prompt diagnosis of CIED infection allows for early management with antibiotics and device removal, which is typically needed for resolution of the infection.
- Prevention of CIED infection is an important strategy, and more research is needed to inform the incidence of CIED infection, risk factors, and devices and techniques to minimize the risk of infection.
DIAGNOSIS
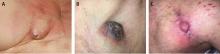
Prompt and accurate diagnosis of CIED infection is critical as it allows for early management with antibiotic therapy and device removal. As the number of CIED implantations increases, providers on the front lines—emergency, family practice, and internal medicine physicians—will play an increasing role in recognizing and diagnosing CIED infection. Patients with CIED infection present with a range of signs and symptoms including fever, chills, erythema, swelling, drainage, tenderness, malaise, erosion, and warmth of the skin overlying the generator pocket.2 In 55% of cases, patients present with localized pocket infection, while the remaining patients have signs of an endovascular infection without obvious pocket involvement.12 Localized pocket infection is more common during the first year after device implantation. CIED-associated endovascular infections occur more commonly in patients with multiple comorbidities including diabetes, renal failure, prior heart valve operation, rheumatic heart disease, and prior bloodstream infection.2 Despite the theoretical divide in CIED infections (endovascular vs pocket), overlap is common: many patients with pocket infection show evidence of bacteremia and vegetations on the leads.
Diagnosing pocket infection from the physical examination can be difficult due to the often subtle manifestations of the underlying pathophysiology and because visible changes to the pocket can occur over weeks and months. Furthermore, differentiating superficial infection, hematoma, seroma, and allergic reactions from deep pocket infection can be challenging. In cases when the diagnosis is not clear and there are no systemic findings of infection, conservative management with close follow-up is reasonable. Similarly, the diagnosis of endovascular infection is sometimes delayed because the symptoms are not very specific or because of a lack of awareness of the presence of a CIED and its role in endovascular infection.
MANAGEMENT
A multidisciplinary approach involving cardiology, infectious disease, electrophysiology, and cardiothoracic surgery teams is required to optimize outcomes in patients with CIED infection. CIED infection is particularly difficult to treat with antibiotic therapy alone because it involves infection of an implanted device and an associated biofilm that is resistant to the effects of antibiotics. Once infection is confirmed, antibiotic therapy serves as an adjunct to the complete removal of the hardware. Most patients receive 2 weeks of intravenous antibiotics after removal of an infected CIED, with longer courses for patients with Staphylococcus aureus infection or documented endocarditis.21
Infectious disease consultation is paramount in order to choose the appropriate type and duration of antibiotic therapy. Conservative approaches that involve using antibiotics alone or incomplete system removal have high failure rates with high rates of morbidity and mortality.13,21–28 However, chronic antibiotic suppressive therapy may be considered as a palliative measure for patients who are not candidates for lead extraction.
DEVICE REMOVAL
Confirmation of CIED infection is a class I indication for device removal and the patient should be referred to an electrophysiologist. Transvenous lead extraction (TLE) is a percutaneous procedure performed by the electrophysiologist in the electrophysiology laboratory or hybrid operating room with cardiothoracic surgery support, and it is generally performed under general anesthesia with invasive hemodynamic monitoring. After opening and debriding the infected pocket, the generator is disconnected from the leads. After the lead tips are unscrewed from the myocardium, gentle traction is applied to determine if the leads can easily be removed. If traction is unsuccessful, additional tools (both powered or mechanical sheaths) are used to complete the lead extraction29; the goal is to lyse and free the fibrotic attachments between parallel leads and between the leads and vessel wall or the myocardium. Once the lead is freed from the adhesions it can be removed safely.