News

Method identifies effective treatments for leukemias, lymphomas
- Author:
- HT Staff
Publish date: November 22, 2017
News
PTSD can persist in cancer survivors
- Author:
- HT Staff
Publish date: November 22, 2017
News

Withdrawn drug receives orphan designation for HA
- Author:
- HT Staff
Publish date: November 22, 2017
News
Nilotinib approved to treat kids with CML in EU
- Author:
- HT Staff
Publish date: November 21, 2017
News
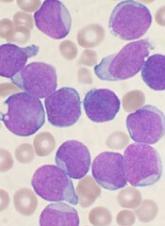
CD22-CAR therapy shows activity in rel/ref B-ALL
- Author:
- HT Staff
Publish date: November 21, 2017
News

NOACs may do less harm to kidneys than warfarin
- Author:
- HT Staff
Publish date: November 21, 2017
News
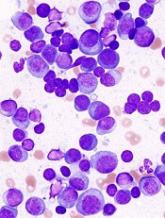
Technique may predict treatment outcomes in MM
- Author:
- HT Staff
Publish date: November 20, 2017
News

Antimalarial might fight Zika virus
- Author:
- HT Staff
Publish date: November 19, 2017
News

Team discovers mechanism of resistance in AML
- Author:
- HT Staff
Publish date: November 18, 2017
News
Product approved to treat patients with hemophilia A and inhibitors
- Author:
- HT Staff
Publish date: November 17, 2017
News
FDA expands approval of obinutuzumab
- Author:
- HT Staff
Publish date: November 17, 2017
News
CAR T-cell therapy on fast track with FDA, EMA
- Author:
- HT Staff
Publish date: November 17, 2017
News
Rigosertib produces better OS in MDS than tAML
- Author:
- HT Staff
Publish date: November 16, 2017
News

Drug can treat severely ill SCD patients, case suggests
- Author:
- HT Staff
Publish date: November 16, 2017
News

FDA approves generic clofarabine
- Author:
- HT Staff
Publish date: November 16, 2017