Tedizolid Use in Immunocompromised Patients
Immunocompromised patients are often susceptible to opportunistic infections, including those caused by multidrug-resistant organisms (MDROs). Transplant recipients are at high risk for developing infections due to lifelong immunosuppressive therapy.1,2 Additionally, patients receiving chemotherapy and those with HIV and AIDS are in an immunocompromised state.3-8
Regardless of the etiology for immunosuppression, decreased absolute neutrophil and platelet counts are seen in this condition. Although immunosuppressed individuals may be at increased risk of Gram-negative or Gram-positive infections, this review focuses on the treatment of Gram-positive bacterial infections. Of particular concern are opportunistic infections caused by Gram-positive MDROs, including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus species (VRE), penicillin-resistant Streptococcus pneumoniae, and Nocardia species. Treatment of infections in the immunocompromised patient population warrants careful antimicrobial selection to ensure that a patient’s immune system is not further compromised due to adverse effects (AEs) secondary to therapy. As such, clinicians are exploring alternative antimicrobials, such as tedizolid, to treat various opportunistic infections.
Recently, requests at the William S. Middleton Memorial Veterans Hospital in Madison, Wisconsin, for off-label use of tedizolid have increased despite having other cheaper alternatives with comparable Gram-positive coverage. This review examines available literature regarding off-label use of tedizolid with a focus on use in immunocompromised patients.
Tedizolid phosphate (Sivextro) is an oxazolidinone antibiotic prodrug that joined linezolid as the second in its class in 2014. Oxazolidinones inhibit bacterial protein synthesis by binding to the 50S subunit of bacterial ribosomes in Gram-positive bacteria and are often used to treat MRSA and VRE infections.9 In vitro, oxazolidinones have shown bacteriostatic activity against Enterococcus and Staphylococcus species while exhibiting bactericidal activity against most Streptococcus species.10 Tedizolid has a US Food and Drug Administration (FDA) -approved, simplified dosing profile of 200 mg daily for 6 days compared with linezolid 400 to 600 mg twice daily for 10 to 14 days. Both medications are highly bioavailable with direct IV to oral conversion.11,12 Potential, expanded use of tedizolid against Gram-positive MDROs rests on a more favorable AE profile than does its linezolid predecessor. Tedizolid has been associated with less antibiotic-induced myelosuppression, which could prove valuable for immunocompromised patients.13
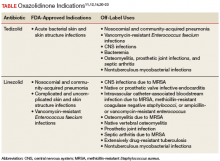
Tedizolid is approved for the sole indication of acute bacterial skin and skin structure infections (ABSSSI), whereas its predecessor has many approved indications and has been used extensively for off-label indications (Table).
Adverse reactions, as determined by 2 phase 2 and 2 phase 3 clinical trials evaluating 1,050 patients treated with tedizolid and 662 patients treated with linezolid, were similar between the oxazolidinones. Nausea was the most common AE and was reported in 8% and 12% of patients taking tedizolid and linezolid, respectively. Other common AEs (1%-6%) reported for both agents included vomiting, diarrhea, headache, and dizziness.11 Myelosuppression, peripheral neuropathy, and optic nerve disorders were the most common severe AEs reported with oxazolidinones. Tedizolid demonstrated a significantly decreased incidence of neutropenia (3%), defined by absolute neutrophil count 9/L compared with that of linezolid (7%) (P = .024).13 Evaluation of peripheral neuropathy and optic nerve disorders within the tedizolid and linezolid groups revealed similar incidences (peripheral neuropathy 1.2% vs 0.6%; optic nerve disorders 0.3% vs 0.2%, respectively).11
There is one preclinical trial that described the use of tedizolid in a murine model. A murine model study compared the antistaphylococcal killing effect of doses of tedizolid equivalent to human exposures ranging from 200 to 3,200 mg/d in both granulocytopenic and normal mice. The mice were evaluated at 24, 48, and 72 hours after therapy initiation. The presence of granulocytes had a dramatic effect on the antimicrobial effect of tedizolid. Dose response, demonstrated by the ratio of the area under the curve over the minimum inhibitory concentration, was on average > 25-fold for nonneutropenic vs neutropenic models. Near maximal effect of the nonneutropenic group, irrespective of duration of therapy, was achieved at the lowest dose tested (an exposure of about 200-mg tedizolid phosphate per day in humans).This study suggests that immunocompromised patients may warrant higher doses of tedizolid than the currently FDA-approved dose due to a decreased number of granulocytes available for modulating bacterial infections.15
Use of tedizolid doses higher than that which is FDA-approved may negate the favorable AE profile. A phase 1 clinical study was conducted to evaluate the safety, tolerability, and pharmacokinetics of tedizolid compared with those of linezolid in 40 healthy volunteers in a 21-day multiple ascending dose study.16 Subjects were stratified into 5 treatment cohorts: 200-, 300-, or 400-mg tedizolid orally once a day, 600-mg linezolid orally twice a day, and placebo. Tedizolid given at 200 mg had a hematologic safety profile similar to that of placebo. However, mean platelet counts decreased over time in a dose-dependent manner for tedizolid, with the 400-mg tedizolid and linezolid groups reporting similar reductions in platelet counts.16
Some evidence is available examining linezolid in neutropenic patients. Rafailidis and colleagues reviewed available literature regarding linezolid in neutropenic patients with Gram-positive infections. Evaluation of linezolid administration at usual doses to 438 neutropenic patients from 2 prospective comparative studies, a prospective cohort study, 2 retrospective studies, and 8 case reports was performed. Results of the evaluation revealed a clinical cure rate between 57% and 87% in the intention-to-treat population of the prospective studies.17 Given the similarities in bacterial spectrum of activity between linezolid and tedizolid, it may be reasonable to infer that tedizolid’s decreased myelosuppression profile would make it useful in the setting of neutropenia in immunocompromised patients.
There is little evidence regarding the use of tedizolid in immunocompromised patients, as only 2 case reports were found. The first described a 60-year-old male postrenal transplant complicated with VRE bacteremia, rhabdomyolysis, and thrombocytopenia. This patient was treated with prolonged tedizolid 200 mg daily due to multiple contraindications for treatment with other antibiotics. The patient was cured with a 14-day course of tedizolid without any noted AEs.18
The second identified case report described the use of tedizolid for the treatment of central nervous system (CNS) manifestations secondary to nocardiosis. Effective treatment of CNS nocardiosis requires high concentrations and prolonged duration of antimicrobial exposure. This case report described a 68-year-old, chronically immunocompromised female patient with multiple myeloma who was hospitalized for 3 months for the treatment of a CNS nocardiosis infection. After discharge, the patient was treated with an oral regimen of 200-mg tedizolid daily in combination with sulfamethoxazole/trimethoprim (800 mg/160 mg) 3 times daily. After 6 months of combination therapy, magnetic resonance imaging revealed complete resolution of nocardiosis-related central lesions. Although the patient’s malignancy advanced during combination antibiotic therapy, the patient’s absolute neutrophil count remained stable and showed an increase in absolute CD4+ cell counts with no other documented AEs.19
Tedizolid is the latest FDA-approved oxazolidinone antibiotic for susceptible Gram-positive acute bacterial skin and skin structure infections. It has a simplified and shorter duration of treatment and imparts similar AEs at improved rates compared with that of linezolid, most notably in relation to hematologic AEs. Due to the lack of established literature and an agreed-upon dosing strategy for the use of tedizolid in immunocompromised patients, tedizolid therapy for Gram-positive infections in immunocompromised patients should be reserved for salvage therapy when more established Gram-positive antibiotic agents lack efficacy or when patient contraindications to their use exist.