What is your diagnosis?
Hodgkins lymphoma of the liver
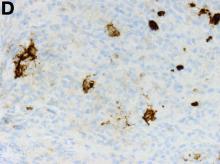
The gallbladder (Figure B) as well as the intraoperative liver biopsy (Figure C; insert showing cells under higher power) showed non-necrotizing granulomas along with scattered infiltration by atypical large cells morphologically consistent with Hodgkin-Reed-Sternberg cells in a lymphoid background (Figures B, C, green arrows). Immunohistochemistry showed these were positive for CD30 (Figure D, liver biopsy), weakly positive for PAX5, and negative for CD15, CD20, CD79a, and ALK-1. Given the pathologic findings, the patient was diagnosed with Hodgkins lymphoma.
The patient had a history of mediastinoscopy and lymph node biopsy in the past at an outside hospital with reported noncaseating granulomas and no other abnormalities; those slides could not be obtained for independent review. Primary lymphomas of the liver are exceedingly rare, but advanced lymphoma can have liver involvement.1 Hodgkins lymphoma of the liver is extremely uncommon.2 It can present with fever, hepatomegaly, and jaundice.1 The diagnostic yield of a liver biopsy ranges from 5% to 10% depending on core versus wedge biopsy.1 Pathologically, there is portal inflammation and atypical histiocytic aggregates but Hodgkin-Reed-Sternberg cells are required for diagnosis. These cells stain positive for CD15 and CD30 in around 80% of cases.3 Lymphoma should remain in the differential when granulomas are seen in the liver biopsy. Our patient clinically decompensated by the time the diagnosis was confirmed. The family decided not to pursue aggressive treatment in hospital and the patient was discharged home where she expired.
References
1. in: R.N.M. MacSween (Ed.) Pathology of the liver. Edinburgh: Churchill Livingstone. ; 1979
2. Levitan R, Diamond H, Lloyd C. The liver in Hodgkin’s disease. Gut. 1961;2:60.
3. Kanel GC, Korula J. Atlas of liver pathology. Elsevier/Saunders, Philadelphia; 2005.